You can easily make Ice cube Tray Vinegar Battery at Home. The Hypothesis and step by step experiment procedure is explained in this post.
Aim of the Project
To make a battery using simple house hold items such as Ice tray, Nails etc.
Materials required

Fig. 1 – Materials Required to Make Ice Cube Tray Vinegar Battery
- Ice trays
- Nails
- Copper-wire
- Vinegar / Salty water
- LED
Hypothesis behind Ice Cube Tray Vinegar Battery
The working principle of our Ice Cube Tray Vinegar Battery is very simple. Whenever two different metallic substances like Zinc and copper are dipped in an acidic medium (electrolyte), then a chemical reaction happens between the metals and the electrolyte which further generates a charge. If you want to generate a larger charge, then connect few similar cells together in series.
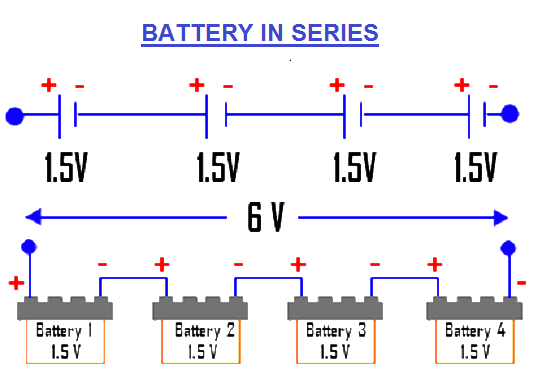
Fig. 2 – Battery in series to generate a larger charge
Explanation of Ice Cube Tray Vinegar Battery Experiment
Ice Cube Tray Vinegar Battery consists of two metals i.e. Zinc and Copper dipped in an acidic medium. In our experiment, the acidic medium is Vinegar.
Zinc is available in the galvanization of the nail and Copper is in the wire. As explained earlier , the acid comes from the Vinegar. To be very specific, Vinegar contains 92-96% water and 4-8% acetic acid. It is the acetic acid which actually provides the acidic medium.
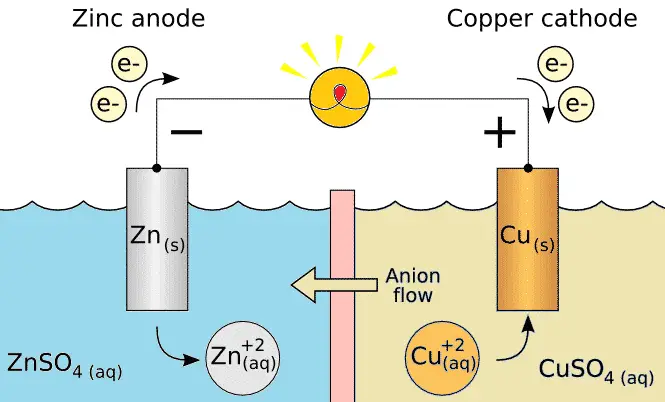
Fig. 3 – Flow of Electrons in the Ice Cube Tray Vinegar Battery
Both the metals acts as electrodes of the battery. It is these electrodes through which the electrons move in and out of Battery when the circuit is completed. This causes an electron flow to start in the circuit and so a current is generated. The flow of current is from Wire (Copper) through the Vinegar to the Nail (Zinc).
Once your Ice Cube tray Vinegar Battery is ready, the LED is placed in the Ice Cube Battery Slot to complete the circuit. The current flowing through the wire will pass through the LED and excite the atoms in it. As the atoms excite, they will emit a particular color wave-length and so the LED will start glowing.
An LED requires a particular direction of electron flow to glow. It is therefore, if your LED does not glow initially, then interchange the foots of the LED and correct the electron flow. You LED will start glowing.
How to make an Ice Cube Tray Vinegar Battery Step by Step
The step by step process to make an Ice Cube Tray Vinegar Battery is as follows: –
Step 1
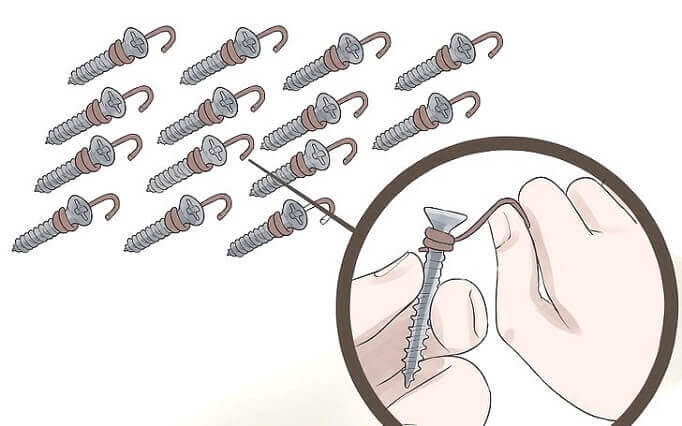
Fig. 4 – How to Wrap Copper Wire on the Nails
Tightly wrap all nails with Copper wire one by one. Just keep it in mind that you have to make 2-3 turns of copper wire on each nail near to the its tip. Try to keep sufficient tail length of the nail free as it has to dip in the acidic solution.
Step 2

Fig. 5 – Placing Nails in the Ice Tray
Place all the nails one by one in each slot of Ice cube tray. Remember that you have to place the nails such that if nail is in one slot, then the end of the copper wire turned with the nail goes to the next slot.
Ensure that the copper wire attached to a nail should not touch the wire or nail of another slot of Ice Cube Tray.
Step 3
Now fill each slot of Ice Cube Tray with Distilled White Vinegar.

Fig. 6 – Pouring Vinegar in Ice Cube Tray
Step 4
Place one “foot†of LED in the Ice Cube Tray slot with only copper wire inside it. Similarly, place another “foot” of the LED in the slot having only nail in it.
If the LED glows, your experiment is complete and ready for demonstration. If the LED does not glow, then just interchange the foots of the LED dipped in the slots. Your LED will start glowing.

Fig. 7 – LED placement in Ice Cube Tray Battery
Recommendations for the Ice Cube Tray Vinegar Battery Experiment
- Always use Vinegar having more acidic content.
- If you want larger current, then use more Ice Cube Trays in series.
- Disconnect the circuit when not in use by removing LED. This Ice Cube Tray Vinegar Battery will be strong initially and the potential difference will slightly decrease with use. So If using it for a project, then connect the LED only when you want to show the result.
Read more about other Alternative Energy Storage Methods.
