pH Measurement specifies the degree of relative acidity or alkalinity of an aqueous solution at a given temperature. It is generally measured using a pH Meter. All living beings depend on a proper pH level to sustain life and hence pH Measurement becomes an important aspect of our lives. This post will discuss in detail about what is pH Measurement, its working principle, how it is measured, its applications, advantages and disadvantages.
What is pH Measurement
pH Measurement specifies the degree of relative acidity or alkalinity of an aqueous solution at a given temperature. Theoretically, pH is derived from the word “Pondus Hydrogenii” which means “Potential Hydrogen†or Power of Hydrogen.
Fig. 1 – Different Types of pH Meters
pH is represented in the form of an equation as the negative logarithm of the Hydrogen ion concentration.
In an aqueous solution, a pH of 7 describes a neutral solution because the activities of Hydrogen (H+) and Hydroxide ions (OH-) are equal. The solution is described as acidic when the pH is below 7,as the activity of Hydrogen ion is dominant when compared to Hydroxide ion. Hence a solution is termed more acidic when the Hydrogen ion activity increases and the pH value decreases.
On the contrary, the solution is described as basic (or alkaline) when the pH is above 7, as the activity of Hydroxide ion is dominant when compared to Hydrogen ion. Fig. 2 below shows the pH scale of different products that we use every day. A change in one unit of pH represents a tenfold change in Hydrogen ion concentration.
Fig. 2 – The pH Scale of Common Products
How is pH Measurement Done
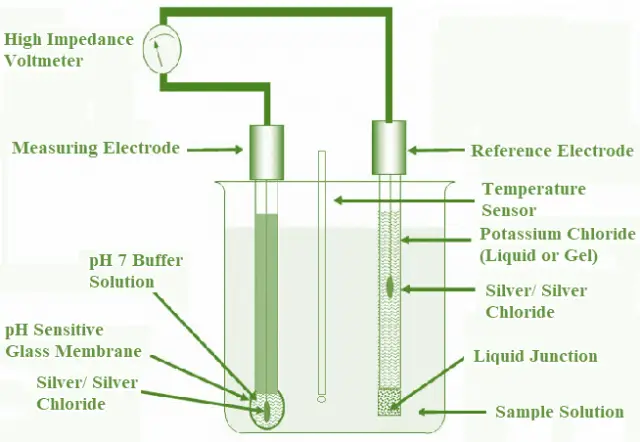
Generally, pH Measurement is obtained precisely using a pH Meter. It has few key components namely Measuring Electrode, Reference Electrode, Temperature Sensor and the Sample Solution being measured. The pH Meter measures the voltage of an electro chemical cell and based on the Temperature Sensor determines the pH of a solution. Off late, in most of the pH Meters the electrodes and the Temperature Sensor are fabricated into a single body and are called as Combination Electrodes. Fig. 3 shows pH Measurement using Measuring Electrode made of glass, Reference Electrode, Temperature Sensor and Liquid junction.
The overall potential or the voltage is the algebraic sum of the potentials of the Measuring Electrode, Reference Electrode and the Liquid Junction. The Reference Electrode provides a stable voltage as it has a fixed concentration of Potassium Chloride solution which is a neutral solution. On the contrary, the potential of the Measuring Electrode depends only on the pH of the solution. The potential difference (voltage) between a glass membrane of Measuring Electrode and a Reference Electrode which is dipped in the Sample Solution to be tested is measured.
When the two Electrodes are dipped in the Sample Solution, ion-exchange process occurs where in some of the Hydrogen ions move towards the outer surface of the Measuring Electrode and replace some of the metal ions inside it. Similarly, some of the metal ions move from the Glass Electrode into the Sample Solution. The sensitivity of the Reference Electrode potential to changes in pH is negligible or it is unaffected by changes in pH and hence provides a stable voltage.
Fig. 3 – pH Measurement Using Two Electrode
Ion-exchange process also takes place on the inner surface of the Glass Electrode from the sample solution. This creates a potential difference (Hydrogen- ion activity) between them. The Liquid Junction potential is usually small and relatively constant which mainly depends on the concentration of the ions in the sample solution. All three potentials are summed up and measured by High Impedance Voltmeter.
The potential voltage developed across the Glass Electrode membrane is temperature dependent, with a temperature coefficient of approximately 0.3% per °C. The pH Meters have provisions to correct the pH Measurements as the temperature changes and it is called as Automatic Temperature Compensation (ATC).
The output of the Impedance Voltmeter is Voltage readings and it has to be calibrated to get precise pH Measurement. Calibration is done by dipping the Measuring Electrode into Buffer Solution of known pH which helps in interpreting millivolt reading as pH measurement of the Sample Solution at the given temperature.
Applications of pH Measurement
The applications include:
- pH Measurement is very crucial in Agriculture industry for soil evaluation. Major crops require alkaline environment and hence pH Measurement becomes necessary.
- It is also used in Food industry especially for dairy products like cheese, curds, yogurts, etc.
- It becomes mandatory for chemical and pharmaceutical industries.
- It becomes a significant factor in the production of detergents.
- pH level monitoring is essential in water treatment plants and RO water purifiers.
Advantages of pH Measurement
The advantages are:
- pH Measurement is inexpensive and robust.
- Pocket size pH Meters are user friendly.
- Readings are accurate and precise.
Disadvantages of pH Measurement
The disadvantages are:
- Temperature impacts the output readings.
- pH Measurement using glass electrodes must be clean as deposition on the electrodes affects the readings.
Also Read: How to Identify Bearings by Bearing Number - Calculation and Nomenclature Step Down Transformer - Working Principle, Equation, Types, Advantages & Disadvantages Unijunction Transistor (UJT) - Construction, Working, Characteristics Curve & Applications