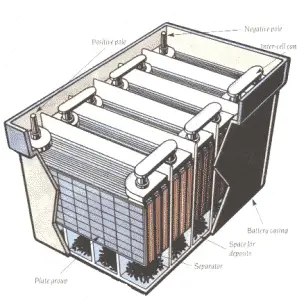
A Battery is an electrochemical system that converts chemical energy into electrical energy. Battery-Cells are the most basic individual component of a battery.
In another words, it can also be defined as :
Battery is an electrochemical device that converts chemical energy into electricity by using a galvanic cell.
We mentioned Galvanic cell. Do you know what is it? A Galvanic cell is a simple device consisting of two electrodes of unlike metals or its compounds (anode and cathode) and an electrolyte (usually acid/alkaline) solution.
I want to clear an ambiguity here. Batteries do not make electricity, they actually store it. It is therefore we can say that a battery is an electrical storage device.
When there is chemical reaction in the 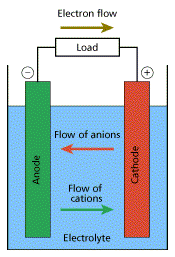 battery, electrical energy is either supplied or stored. In Dry cells, this chemical reaction happens only for a single life. Whereas in rechargeable batteries, this chemical reaction can be repeated many times.
battery, electrical energy is either supplied or stored. In Dry cells, this chemical reaction happens only for a single life. Whereas in rechargeable batteries, this chemical reaction can be repeated many times.
Based on the nature of reversal of chemical reaction, it can be categorized as :
- Primary cell, and
- Secondary cell.
Primary Cell :
When the battery’s chemical reaction is irreversible, it is called a primary cell. In a primary cell, the chemical reaction eats away one of the electrodes, usually the negative electrode. When this happens, the electrode must be replaced or the cell must be discarded.
Secondary Cell :
When the battery’s chemical reaction is reversible, it is called a secondary cell. In a secondary cell, the electrodes and electrolyte are altered by the chemical action. This action takes place when the cell delivers current. A secondary cell may be restored to its original condition by forcing an electric current through it in the direction opposite to that of discharge.
If you liked the post, then share with others and spread the power of knowledge. We will meet again with some interesting topics. Till then, keep learning.



5bk8tu
2f2kin
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.
fantastic issues altogether, you just won a new reader. What may you recommend in regards to your put up that you made a few days in the past? Any positive?
I dugg some of you post as I thought they were very helpful very helpful
Absolutely composed written content, regards for selective information. “You can do very little with faith, but you can do nothing without it.” by Samuel Butler.
When I originally commented I clicked the -Notify me when new comments are added- checkbox and now each time a comment is added I get four emails with the same comment. Is there any way you can remove me from that service? Thanks!
It¦s actually a cool and helpful piece of info. I am satisfied that you simply shared this useful info with us. Please keep us informed like this. Thanks for sharing.
Right now it looks like BlogEngine is the best blogging platform out there right now. (from what I’ve read) Is that what you’re using on your blog?
I have recently started a blog, the information you provide on this web site has helped me greatly. Thank you for all of your time & work. “The inner fire is the most important thing mankind possesses.” by Edith Sodergran.
Thanks , I’ve recently been looking for info about this topic for ages and yours is the greatest I have discovered till now. But, what about the conclusion? Are you sure about the source?
Thank you, I have recently been looking for info about this topic for ages and yours is the best I have discovered till now. But, what about the conclusion? Are you sure about the source?
Keep working ,splendid job!
You have remarked very interesting points! ps decent website . “In music the passions enjoy themselves.” by Friedrich Wilhelm Nietzsche.
Hey very nice site!! Man .. Excellent .. Wonderful .. I will bookmark your site and take the feeds also…I am happy to find numerous helpful info here within the put up, we’d like work out more techniques on this regard, thanks for sharing.
Excellent post. I was checking continuously this blog and I’m impressed! Very helpful info specially the last part 🙂 I care for such information much. I was seeking this certain information for a long time. Thank you and good luck.
It’s the best time to make some plans for the long run and it’s time to be happy. I have learn this submit and if I could I desire to suggest you some attention-grabbing issues or tips. Maybe you can write next articles relating to this article. I want to read more issues about it!
I just couldn’t depart your website before suggesting that I extremely enjoyed the standard info a person provide for your visitors? Is going to be back often in order to check up on new posts
Solid analysis
It¦s in reality a great and helpful piece of info. I¦m glad that you shared this useful information with us. Please keep us up to date like this. Thanks for sharing.
Can I just say what a relief to find someone who actually knows what theyre talking about on the internet. You definitely know how to bring an issue to light and make it important. More people need to read this and understand this side of the story. I cant believe youre not more popular because you definitely have the gift.
Hello there, You’ve done an excellent job. I will certainly digg it and personally recommend to my friends. I’m sure they will be benefited from this website.
you have a great blog here! would you like to make some invite posts on my blog?
Impressive write-up
Those are yours alright! . We at least need to get these people stealing images to start blogging! They probably just did a image search and grabbed them. They look good though!
I am very happy to read this. This is the type of manual that needs to be given and not the accidental misinformation that’s at the other blogs. Appreciate your sharing this greatest doc.
Today, I went to the beach with my kids. I found a sea shell and gave it to my 4 year old daughter and said “You can hear the ocean if you put this to your ear.” She placed the shell to her ear and screamed. There was a hermit crab inside and it pinched her ear. She never wants to go back! LoL I know this is entirely off topic but I had to tell someone!
Good info. Lucky me I reach on your website by accident, I bookmarked it.
The subsequent time I learn a blog, I hope that it doesnt disappoint me as much as this one. I mean, I do know it was my choice to learn, but I actually thought youd have one thing interesting to say. All I hear is a bunch of whining about something that you may fix for those who werent too busy looking for attention.
I haven¦t checked in here for some time as I thought it was getting boring, but the last few posts are good quality so I guess I¦ll add you back to my daily bloglist. You deserve it my friend 🙂
cpbl粉絲必備的官方認證資訊平台,24小時不間斷提供官方cpbl新聞、球員數據分析,以及專業的比賽預測。
Some really nice and utilitarian information on this site, as well I conceive the style has got fantastic features.
Hello There. I found your blog using msn. This is a really well written article. I’ll make sure to bookmark it and come back to read more of your useful info. Thanks for the post. I will certainly comeback.
Regards for helping out, superb information. “A man will fight harder for his interests than for his rights.” by Napoleon Bonaparte.
Hello very cool blog!! Guy .. Beautiful .. Amazing .. I will bookmark your blog and take the feeds also…I am glad to find a lot of useful information here in the publish, we’d like develop extra strategies on this regard, thanks for sharing.
I really enjoy reading on this website , it holds superb content.
austin hike and bike trail [url=https://otvetnow.ru]https://otvetnow.ru[/url] retail location analytics
Real money pokies Australia – best sites to play and win
This web site is really a walk-through for all of the info you wanted about this and didn’t know who to ask. Glimpse here, and you’ll definitely discover it.
Great line up. We will be linking to this great article on our site. Keep up the good writing.
Hi , I do believe this is an excellent blog. I stumbled upon it on Yahoo , i will come back once again. Money and freedom is the best way to change, may you be rich and help other people.
Great line up. We will be linking to this great article on our site. Keep up the good writing.
Great post, I conceive people should larn a lot from this web site its really user friendly.
Interesting blog! Is your theme custom made or did you download it from somewhere? A theme like yours with a few simple tweeks would really make my blog shine. Please let me know where you got your theme. Cheers
mftmcq
Does your blog have a contact page? I’m having trouble locating it but, I’d like to shoot you an email. I’ve got some creative ideas for your blog you might be interested in hearing. Either way, great blog and I look forward to seeing it grow over time.
I loved as much as you will receive carried out right here. The sketch is tasteful, your authored material stylish. nonetheless, you command get bought an nervousness over that you wish be delivering the following. unwell unquestionably come further formerly again since exactly the same nearly very often inside case you shield this increase.
Thanks for your post. I would really like to say that the cost of car insurance will vary from one plan to another, due to the fact there are so many different facets which bring about the overall cost. By way of example, the brand name of the motor vehicle will have an enormous bearing on the purchase price. A reliable outdated family car or truck will have an inexpensive premium than a flashy expensive car.