Galvanic corrosion may occur in fresh or salt water. It generally happens whenever two/or more metallic elements having unlike galvanic-voltages connects to each other electrically in the presence of an electrolyte.
In other words, it can also be said that Galvanic corrosion is an outcome of the usual property of an active metal (greater in comparison to the other metal) to shred the electrons to the lesser active metallic counterpart. The corrosion increases speedily whenever unlike metallic elements connect to each other electrically. In more layman terms, we can say that a corrosion which happens due to the association of two/or more unlike metals is known as Galvanic Corrosion.
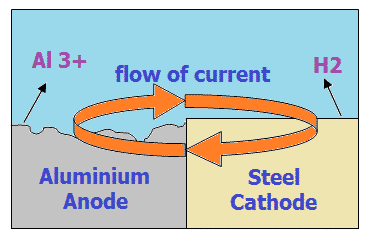 In the figure beside, The aluminium is having the loss of metal in terms of aluminium ion. The flow of electrons is from aluminium to the Steel via an electric medium.
In the figure beside, The aluminium is having the loss of metal in terms of aluminium ion. The flow of electrons is from aluminium to the Steel via an electric medium.
The galvanic corrosion can only happen only in the presence of electric circuitry. With this statement, we can understand that for a galvanic corrosion to happen, it is mandatory to have two or more metals and an electrolytic medium for providing a path for the flow of electrons. In addition, the chemical reaction near the Cathodic material needs the availability of O2 in the electrolytic medium.
Lets understand What is ‘Anode’ and ‘Cathode’ now.
Anode :
The metallic element which loses (donate) the electrons when current flows is known as Anode. It has a ‘-ve’ galvanic voltage in comparison to another metal and so the rate of Corrosion is also more in that.
Cathode :
The another metallic element which accepts the electrons when current flows is known as Cathode. It has ‘+ve’ galvanic voltage in comparison to another metal and so the rate of Corrosion is less in that.
In order to stop the occurrence of Galvanic corrosion, we should at least maintain any one of the below mentioned three conditions :
- The galvanic voltage level of both the metals should be same (before coupling)
- The electric circuit should not be made.
- Presence of electrolytic medium to be removed.
By design a ship is made of steel and the natural tendency of the ships hull is to become anodic with reference to the nonferrous metals and the impurities present within steel. Therefore the ships hull gets corroded.
For oxidation corrosion, visit https://electricalfundablog.com/blog/oxidation-corrosion/708/ . Share if you like the post.

I’m often to blogging and i actually appreciate your content. The article has really peaks my interest. I’m going to bookmark your website and hold checking for new information.
oc7z9f
We are a gaggle of volunteers and starting a new scheme in our community. Your web site provided us with useful information to paintings on. You’ve performed a formidable activity and our entire group will be thankful to you.
umtxae
Appreciating the hard work you put into your blog and detailed information you offer. It’s good to come across a blog every once in a while that isn’t the same old rehashed material. Great read! I’ve saved your site and I’m including your RSS feeds to my Google account.
I believe you have mentioned some very interesting points, appreciate it for the post.
But wanna say that this is extremely helpful, Thanks for taking your time to write this.
I am perpetually thought about this, regards for putting up.
I’ve recently started a web site, the info you offer on this site has helped me tremendously. Thanks for all of your time & work. “The achievements of an organization are the results of the combined effort of each individual.” by Vince Lombardi.
I am glad to be one of many visitants on this outstanding website (:, thanks for putting up.
Appreciate it for this post, I am a big fan of this site would like to go on updated.
I conceive you have observed some very interesting points, thanks for the post.
Excellent write-up
Everything is very open and very clear explanation of issues. was truly information. Your website is very useful. Thanks for sharing.
I cling on to listening to the newscast lecture about receiving boundless online grant applications so I have been looking around for the best site to get one. Could you advise me please, where could i find some?
Informative content
I do not even know how I ended up here, but I thought this post was great. I do not know who you are but certainly you’re going to a famous blogger if you aren’t already 😉 Cheers!
Hi, i feel that i saw you visited my blog thus i got here to “return the prefer”.I am trying to to find issues to improve my website!I guess its ok to use a few of your ideas!!
Keep working ,terrific job!
I consider something truly special in this website.
I enjoy forgathering utile information , this post has got me even more info! .
But wanna input that you have a very decent internet site, I like the layout it really stands out.
I’ve read a few good stuff here. Definitely worth bookmarking for revisiting. I surprise how much effort you put to create such a great informative website.
hello!,I really like your writing very a lot! proportion we keep in touch more about your article on AOL? I need a specialist on this area to solve my problem. May be that’s you! Looking ahead to see you.
Write more, thats all I have to say. Literally, it seems as though you relied on the video to make your point. You obviously know what youre talking about, why throw away your intelligence on just posting videos to your site when you could be giving us something informative to read?
Really superb info can be found on weblog. “Compassion for myself is the most powerful healer of them all.” by Theodore Isaac Rubin.
我們提供官方合作夥伴台灣最完整的棒球即時比分相關服務,包含最新官方賽事資訊、數據分析,以及專業賽事預測。
Hmm is anyone else encountering problems with the pictures on this blog loading? I’m trying to find out if its a problem on my end or if it’s the blog. Any responses would be greatly appreciated.
Howdy! This post couldn’t be written any better! Reading through this post reminds me of my good old room mate! He always kept chatting about this. I will forward this page to him. Pretty sure he will have a good read. Thanks for sharing!
Good write-up, I?¦m regular visitor of one?¦s website, maintain up the nice operate, and It is going to be a regular visitor for a long time.
enterprise antivirus reviews [url=https://otvetnow.ru]https://otvetnow.ru[/url] godaddy secure email login
You have noted very interesting points! ps nice site. “Become addicted to constant and never-ending self improvement.” by Anthony D’Angelo.
I was very pleased to find this web-site.I wanted to thanks for your time for this wonderful read!! I definitely enjoying every little bit of it and I have you bookmarked to check out new stuff you blog post.
Very superb visual appeal on this site, I’d rate it 10 10.
I enjoy the efforts you have put in this, thankyou for all the great articles.
Virtually all of the things you say is supprisingly appropriate and that makes me ponder the reason why I hadn’t looked at this with this light previously. This particular piece truly did turn the light on for me as far as this specific issue goes. However there is actually 1 factor I am not too comfortable with so whilst I make an effort to reconcile that with the central theme of the position, allow me observe what all the rest of your subscribers have to say.Very well done.
Your article helped me a lot, is there any more related content? Thanks!
I have recently started a blog, the information you offer on this web site has helped me greatly. Thank you for all of your time & work.
F*ckin’ amazing things here. I am very glad to see your post. Thanks a lot and i am looking forward to contact you. Will you kindly drop me a mail?
My spouse and I stumbled over here different website and thought I might as well check things out. I like what I see so now i’m following you. Look forward to checking out your web page repeatedly.
UEFA Super Cup live scores, Champions League vs Europa League winners clash
WONDERFUL Post.thanks for share..more wait .. …
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
Thank you for some other informative website. The place else could I am getting that type of info written in such a perfect method? I’ve a mission that I am simply now working on, and I’ve been at the look out for such information.
I believe this is one of the so much significant information for me. And i’m satisfied studying your article. However want to observation on some common things, The website taste is wonderful, the articles is in reality nice :D. Excellent task, cheers
Pretty element of content. I simply stumbled upon your site and in accession capital to claim that I acquire in fact enjoyed account your weblog posts. Any way I will be subscribing in your augment and even I success you get right of entry to consistently rapidly.
Hi there, just become alert to your weblog thru Google, and located that it’s truly informative. I’m gonna be careful for brussels. I’ll appreciate in the event you proceed this in future. Numerous folks will likely be benefited out of your writing. Cheers!