It is generally understood that Corrosion means rust and is treated like an almost universal object of hatred. This word ‘Rust’ has been specifically reserved for iron corrosion. However, Not so many knows that corrosion is such a destructive process which affects almost all metals. Since iron is the first and most extensively used metal by man, there is a possibility that suffering of man due to serious corrosion problems to his precious metal ‘IRON’ must be the reason that this word ‘corrosion’ is mostly connected with iron.
Corrosion of metals remains a serious problem to this day. Solutions are also available. Items can be coated with paint which provide a barrier to attack from the surrounding seawater. However, coatings of such things are normally damaged during handling. It is therefore this may not be a solution to this long-term exposure. An alternate method regularly used in addition with skin coatings is use of Cathodic protection. In this method, the component which is likely to be corroded is fitted with anodes normally in the form of blocks of metallic alloys. Sacrificial corrosion of the anodes provides a form of electrical protection.
Why corrosion happens??
Corrosion can be defined as an electro-chemical reaction that involves metal/metals in particular. A chemical reaction and electrical properties does the entire thing, and finally results in the metal’s partial loss. One of the most familiar example for everyone (including me and you) is the presence of rust in our house hold metal stuffs like utensils.
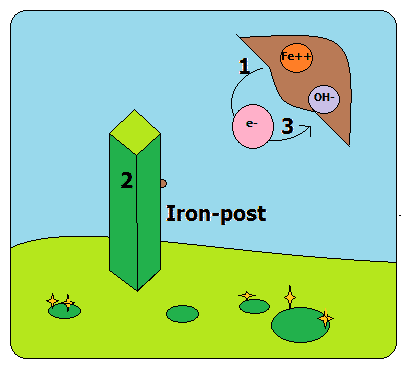
Let us take an example of an iron-post placed in a football-ground. The rain has just stopped and the areas ‘1’ and ‘3’ of the iron-post are damp. In the presence of water (as a medium), there is a movement of electrons in the metallic structure from area 1 to area 3. Thus, in 1st area there is a loss of ‘Fe’ ions through water. Similarly, in 3rd area, just because of the availability of O2 (oxygen) and the accumulation of extra electrons there, the molecules of water combine to O2 and the extra electrons to form hydroxides.
 The corrosion can be classified into two categories. These are :
- Oxidation Corrosion, and
- Galvanic Corrosion.



At this time it sounds like BlogEngine is the top blogging platform available right now. (from what I’ve read) Is that what you’re using on your blog?
I am typically to running a blog and i actually appreciate your content. The article has actually peaks my interest. I’m going to bookmark your web site and preserve checking for brand spanking new information.
Thanks for helping out, wonderful information.
That is the fitting weblog for anybody who desires to search out out about this topic. You realize a lot its almost arduous to argue with you (not that I actually would need…HaHa). You undoubtedly put a brand new spin on a topic thats been written about for years. Nice stuff, simply nice!
This web site is really a walk-through for all of the info you wanted about this and didn’t know who to ask. Glimpse here, and you’ll definitely discover it.
Utterly pent content material, Really enjoyed looking at.
Respect to website author, some excellent information .
Hello there, You’ve performed a fantastic job. I will definitely digg it and personally recommend to my friends. I am sure they’ll be benefited from this website.
Well I definitely liked studying it. This article procured by you is very helpful for proper planning.
Hello, Neat post. There’s a problem together with your website in internet explorer, might test this?K IE nonetheless is the market leader and a good element of people will omit your fantastic writing because of this problem.
Thanks for sharing. I read many of your blog posts, cool, your blog is very good.
Your style is so unique compared to many other people. Thank you for publishing when you have the opportunity,Guess I will just make this bookmarked.2
Good day very cool blog!! Man .. Beautiful .. Amazing .. I’ll bookmark your website and take the feeds also…I’m satisfied to seek out so many useful information right here within the put up, we’d like work out more strategies in this regard, thank you for sharing.
Great work! That is the type of info that should be shared around the net. Shame on the seek engines for now not positioning this submit higher! Come on over and consult with my site . Thanks =)
Hi, Neat post. There is a problem with your website in internet explorer, would test this… IE still is the market leader and a huge portion of people will miss your great writing due to this problem.
Hello, you used to write fantastic, but the last several posts have been kinda boring?K I miss your super writings. Past several posts are just a little out of track! come on!
Hi there! I simply wish to give a huge thumbs up for the great data you’ve got here on this post. I will probably be coming back to your blog for extra soon.
Thank you for the sensible critique. Me & my neighbor were just preparing to do some research on this. We got a grab a book from our area library but I think I learned more from this post. I’m very glad to see such excellent info being shared freely out there.
Greetings! I know this is somewhat off topic but I was wondering if you knew where I could locate a captcha plugin for my comment form? I’m using the same blog platform as yours and I’m having trouble finding one? Thanks a lot!
I believe you have mentioned some very interesting details, appreciate it for the post.
You should take part in a contest for one of the best blogs on the web. I will recommend this site!
Please let me know if you’re looking for a article author for your weblog. You have some really good posts and I believe I would be a good asset. If you ever want to take some of the load off, I’d really like to write some articles for your blog in exchange for a link back to mine. Please send me an e-mail if interested. Cheers!
I simply couldn’t leave your website before suggesting that I extremely loved the standard information an individual provide on your visitors? Is gonna be back often to investigate cross-check new posts
foundations of it service management with itil 2011 [url=https://otvetnow.ru]https://otvetnow.ru[/url] financial assistance for small business
Please let me know if you’re looking for a article writer for your weblog. You have some really great posts and I think I would be a good asset. If you ever want to take some of the load off, I’d really like to write some material for your blog in exchange for a link back to mine. Please send me an e-mail if interested. Regards!
Hello! This post could not be written any better! Reading this post reminds me of my old room mate! He always kept talking about this. I will forward this page to him. Fairly certain he will have a good read. Thank you for sharing!
Wow that was strange. I just wrote an really long comment but after I clicked submit my comment didn’t appear. Grrrr… well I’m not writing all that over again. Anyhow, just wanted to say wonderful blog!
Thank you for another informative blog. Where else could I get that type of information written in such an ideal way? I’ve a project that I am just now working on, and I’ve been on the look out for such information.
hi!,I really like your writing very much! share we be in contact more approximately your post on AOL? I require a specialist on this area to solve my problem. Maybe that is you! Looking forward to peer you.
You could certainly see your skills in the work you write. The world hopes for even more passionate writers like you who aren’t afraid to say how they believe. Always follow your heart.
I love looking through and I conceive this website got some truly useful stuff on it! .
Hello there, I found your website by way of Google even as searching for a similar matter, your web site got here up, it appears great. I have bookmarked it in my google bookmarks.
It?s actually a cool and helpful piece of info. I?m glad that you shared this useful info with us. Please keep us up to date like this. Thanks for sharing.