The fundamental concept of Atom and its structure helps in better understanding of organic molecular structure. This post will discuss in detail about what is an Atom, structure of an Atom (Atomic Structure), different types of atomic models and their applications.
What is an Atom
Scientifically speaking, Atom is the smallest constituent unit of any kind of matter. More simply, Atoms can be called the Legos of the universe. Just like hundreds of different Lego blocks are connected to make a single structure, Atoms too are the very basic building blocks of everything that we can see around us.
Fig. 1 – Introduction to Atom
Democritus, an ancient Greek philosopher, was one of the first atomic theorists who stated that all matter consists of Atoms and they are completely solid.
He also stated that it is the smallest unit of matter which could not be broken down (divided) further. He called his theory as the “Atomic Theory of Universeâ€. This principle was then adopted by John Dalton and today the world recognizes him to be the ‘Father of Modern Atomic Theory’.
However, the theory of Atoms was discovered nearly 2500 years ago by an Indian sage and philosopher named Acharya Kanad. He called the indivisible matter as “Anu†(Sanskrit word) which means Atom. He was a pioneer in Atomic Theory and founded a school called as ‘Vaisheshika School of Philosophy’ where he taught his Aphorisms on “Atomic Theoryâ€.
Fig. 2 – Indian Sage Acharya Kanad
Structure of an Atom
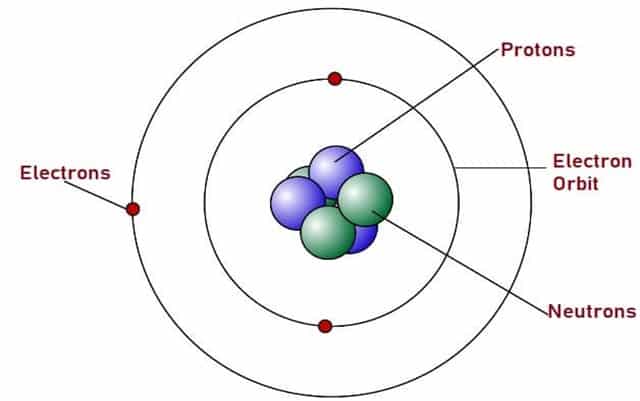
Everything in this universe is made out of Atoms. With a modern understanding of science, we have established certain facts about the nature of Atoms. We know that each Atom is a nucleus made of three basic parts. They are:
- Protons
- Neutrons
- Electrons (Not a part of the Nucleus)
Fig. 4 – Atomic Structure
Protons
A Proton is the positively charged part of the Nucleus and is an essential part of any element’s chemical properties. The number of Protons in an Atom determines the nature of each element. Atoms identity is determined by its atomic number.
Atomic numbers are defined as the number of Protons in the Nucleus of an atom. For E.g. Oxygen (the most essential gas for human survival) has an Atomic number of 8, and in its Nucleus, it has 8 Protons.
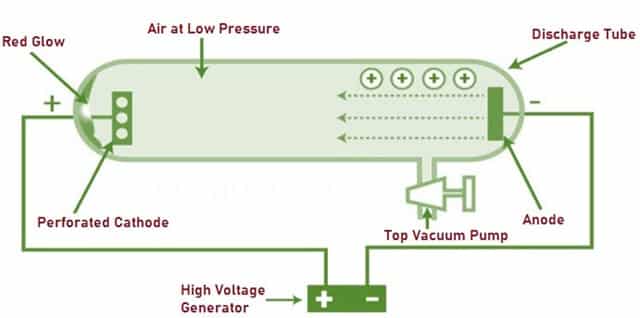
Protons were discovered by Ernest Rutherford in 1919. He did so by using the α-particle bombardment experiment. In this experiment, a vacuum tube is, with the help of a high voltage generator subjected to bombardment with positively charged (α) particles.
Fig. 3 – Rutherford’s Experiment Using α-Particles
On doing this, the scintillation detectors inside the tube detected the presence of Hydrogen Nuclei. After further experimentation, he discovered that the same Hydrogen Nuclei were present inside Nitrogen Atoms. This demonstrated the presence of Hydrogen Nuclei inside a Nitrogen Atom. Hence showing that Hydrogen Nuclei which were then renamed to ‘Proton’, is present inside every Atom.
Neutrons
Neutron is the charge-less constituent subatomic particle of an Atom. A Neutron is also a Nucleon. Inside the Nucleus, both Neutrons and Protons work similarly hence they both have equal masses.
The properties of an Atom are also determined by the number of Neutrons in its Nucleus. Neutrons play a big part in emission of Nuclear energy. During the decades after its discovery, scientists used Neutrons to power a lot of Nuclear experiments.
Electrons
Electron is the negatively charged subatomic particle of an Atom. Electrons revolve around the Nucleus in Orbits. These Orbits are divided into sub-orbits and further into Orbitals. The innermost Orbits have the least energy level and the least number of Electrons.
On moving outwards, the energy level of the Orbitals increases, and the number of Electrons also increases. At these higher energy levels, the Orbits divide into more suborbital; hence allowing more Electrons to be added to that Orbit.
The negative charge on an Electron is the negative of the magnitude of the positive charge on a Proton. And we know that a stable Atom is always neutral in charge. For this charge-less condition to be possible, the number of Protons must be equal to the number of Electrons. Due to this, the total charge on the Atom will be equal to zero.
Types of Atomic Models
Several Atomic Models emerged in the history of Atomic Physics. Few models are listed as follows:
- J.J Thomson’s Atomic Model (Plum Pudding Model)
- Rutherford’s Atomic Model
- Bohr’s Atomic Model
- Quantum Mechanical Atomic Model
J.J Thomson’s Atomic Model (Plum Pudding Model)
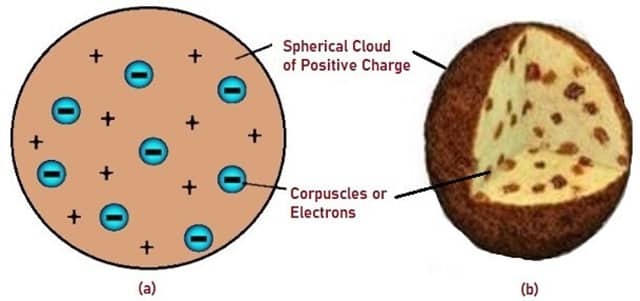
The first model of an Atom was presented by J.J Thomson, he presented the Plum Pudding Model. In this model of the Atom, the positively charged part was spread over the total volume of the Atom, and the electrons seemed to be embedded inside this bowl of positively charged pudding and the electrons were compared to plums. It’s clear to see why his interpretation of the Atomic structure has been compared to a bowl of Plum Pudding.
Fig. 5 – Schematic Representation of Plum Pudding Model
The limitations of this theory were made abundantly clear by Rutherford in 1911, he claimed that Thomson’s model of the Atomic structure was ‘wrong’ on the basis of his experiments that demonstrated how the positively charged nucleus was concentrated at the center of the atom, not spread over the volume of the Atom.
Read More about J.J Thomson’s Atomic Model (Plum Pudding Model) in detail
Rutherford’s Atomic Model
Rutherford proposed the ‘Nuclear Model’ of Atomic Structure. According to this structure, the Nucleus was placed at the center, and Electrons were placed all around the Nucleus. The problem with this model was that the placement of Electrons was not clear.
The Protons and Neutrons were mainly the focus of this model, as they made up for the majority of the mass in the Atom. Whereas, Electrons occupy the most volume.
Fig. 6 – Rutherford’s Atomic Model
The problem with this otherwise sound theory was, as can be seen, the placement of Electrons. This problem would be rectified in a newer model of the Atom in the coming years by Niels Bohr in his Atomic Model.
Bohr’s Atomic Model
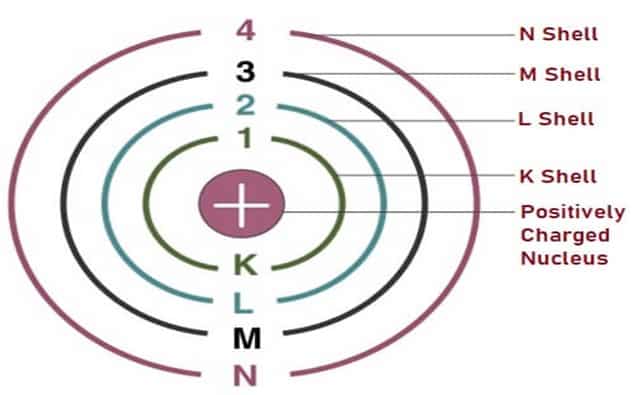
Neil Bohr’s Atomic Model was known as the ‘Shell Model’ in 1913, he proposed the circular orbits of the Electrons and gave the energy level theory. This theory explained how lower energy level orbits hold fewer Electrons as compared to the higher energy level orbits.
This Atomic Model explained the position of Electrons as well as the placement of the Nucleus. It consisted of a dense Nucleus with Electrons revolving around it, just like our solar system.
Fig. 7 – Bohr’s Atomic Model
The only difference between the structure of the solar system and that of Neil Bohr’s atom was that of the force that held it together. The solar system is held together by gravitational forces and the atom is held together by Electrostatic forces of attraction.
Fig. 7 above shows representation of Bohr’s Atomic Model where the energy levels are represented by integers (n=1,2,3,4…) and K, L, M, N Shells are assigned to different orbits. The Electrons move from lower energy level to higher level by gaining energy and vice versa.
Quantum Mechanical Atomic Model
The new ‘Quantum Mechanical Atomic Model’ was presented in 1926 by Erwin Schrodinger. This model of Atoms is very complex and considers the Electrons to be wave-like instead of subatomic particles. He used mathematical equations to calculate the likelihood of the presence of an Electron at one specific point in its orbit.

Fig. 8 – Quantum Mechanical Atomic Model
Since the position of electrons is not clear, the orbits are displayed as clouds rather than perfect paths, because the electron’s position must be determined.
Applications of Atoms
The applications include:
- Based on the Atomic Theory, Nuclear Fission and Fusion experiments were carried out and controlled in Nuclear Plants.
- Atomic Bombs were developed for both war and peace.
- Atomic Theory contributed to revolution in the field of Quantum Physics.
- Study of Atoms helped in the discovery of Transistors, Diodes, CRT (Electron Gun).
Also Read: Random Number Generator – Type, How it Works, Architecture, Application PNP Transistor – Working Principle, Characteristics & Applications Ferroelectric Random Access Memory, FRAM – Cell Structure, Application




knn3jj
8jfwi0
j0k1df
859rml
Thanks for shening. I read many of your blog posts, cool, your blog is very good.
I conceive you have remarked some very interesting details, regards for the post.
It’s a pity you don’t have a donate button! I’d most certainly donate to this excellent blog! I guess for now i’ll settle for bookmarking and adding your RSS feed to my Google account. I look forward to fresh updates and will share this website with my Facebook group. Talk soon!
I like this blog very much, Its a really nice position to read and obtain info .
Hi my loved one! I wish to say that this article is amazing, nice written and come with almost all important infos. I would like to look extra posts like this .
This really answered my problem, thank you!
Hello! Do you know if they make any plugins to protect against hackers? I’m kinda paranoid about losing everything I’ve worked hard on. Any tips?
Great write-up, I’m normal visitor of one’s blog, maintain up the excellent operate, and It’s going to be a regular visitor for a lengthy time.
This blog is definitely rather handy since I’m at the moment creating an internet floral website – although I am only starting out therefore it’s really fairly small, nothing like this site. Can link to a few of the posts here as they are quite. Thanks much. Zoey Olsen
Howdy! This is my first comment here so I just wanted to give a quick shout out and tell you I truly enjoy reading your posts. Can you recommend any other blogs/websites/forums that deal with the same topics? Thank you so much!
I like this post, enjoyed this one thankyou for putting up.
You are my aspiration, I own few blogs and rarely run out from post :). “Yet do I fear thy nature It is too full o’ the milk of human kindness.” by William Shakespeare.
Thank you for sharing excellent informations. Your web-site is so cool. I’m impressed by the details that you?¦ve on this blog. It reveals how nicely you understand this subject. Bookmarked this website page, will come back for extra articles. You, my pal, ROCK! I found simply the information I already searched everywhere and simply couldn’t come across. What a great site.
I¦ve read several good stuff here. Definitely price bookmarking for revisiting. I wonder how much effort you put to make this sort of fantastic informative web site.
Hi there just wanted to give you a quick heads up. The words in your post seem to be running off the screen in Opera. I’m not sure if this is a formatting issue or something to do with internet browser compatibility but I figured I’d post to let you know. The design look great though! Hope you get the issue resolved soon. Cheers
I have recently started a website, the information you provide on this web site has helped me tremendously. Thank you for all of your time & work.
I love your blog.. very nice colors & theme. Did you create this website yourself? Plz reply back as I’m looking to create my own blog and would like to know wheere u got this from. thanks
I love your blog.. very nice colors & theme. Did you create this website yourself? Plz reply back as I’m looking to create my own blog and would like to know wheere u got this from. thanks
Excellent beat ! I wish to apprentice at the same time as you amend your web site, how could i subscribe for a weblog website? The account helped me a applicable deal. I were tiny bit familiar of this your broadcast offered shiny transparent concept
Hey there just wanted to give you a quick heads up. The text in your article seem to be running off the screen in Safari. I’m not sure if this is a format issue or something to do with browser compatibility but I figured I’d post to let you know. The design and style look great though! Hope you get the issue fixed soon. Cheers
I am really loving the theme/design of your site. Do you ever run into any browser compatibility issues? A small number of my blog visitors have complained about my website not operating correctly in Explorer but looks great in Chrome. Do you have any recommendations to help fix this issue?
Write more, thats all I have to say. Literally, it seems as though you relied on the video to make your point. You obviously know what youre talking about, why throw away your intelligence on just posting videos to your blog when you could be giving us something enlightening to read?
wasp control birmingham [url=https://otvetnow.ru]https://otvetnow.ru[/url] nationwide insurance columbia md
Your article helped me a lot, is there any more related content? Thanks!
I like this web site because so much utile stuff on here : D.
I think other web-site proprietors should take this web site as an model, very clean and magnificent user friendly style and design, let alone the content. You are an expert in this topic!
爱一番海外版,专为华人打造的高清视频平台结合大数据AI分析,支持全球加速观看。
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.
I in addition to my friends happened to be studying the excellent techniques on your website then instantly got an awful suspicion I had not expressed respect to the blog owner for those strategies. All the boys appeared to be as a consequence thrilled to see them and have definitely been tapping into those things. We appreciate you genuinely indeed considerate as well as for making a decision on these kinds of superior things millions of individuals are really desperate to know about. Our sincere regret for not saying thanks to you sooner.
I have learn some just right stuff here. Certainly worth bookmarking for revisiting. I surprise how so much attempt you place to create such a fantastic informative website.