Thomson’s Atomic Model also called as ‘Plum Pudding Model’ was the most accepted Atomic Model during the year 1904-1910, which emphasized on the inner structure of the Atom. This post will discuss what is Thomson’s Atomic Model, postulates of J.J. Thomson’s Model, How does Plum Pudding Model Work, applications and limitations.
What is Thomson’s Atomic Model
Thomson’s Atomic Model (also called as ‘Plum Pudding Model‘) explains the inner structure of the atom theoretically. ‘J.J. Thomson’ was a great physicist who has the credit of discovering ‘Electrons’ and was awarded Nobel Prize for his discovery. He conducted his experiment using Cathode Ray Tube (CRT). His theory states that an Atom is made up of thousands of Electrons and an Electron is 2000 times lighter than Proton. He called the ‘Electrons’ as “Corpusclesâ€.

Fig. 1 – Introduction to Thomson’s Atomic Model
Thomson’s Atomic Model states that an Atom is electrically neutral and is made up of ‘Corpuscles’ which act as Negative ions. He also assumed that these Negative ions or Electrons moved in an orbit in a sphere which had equal positive charges. This scientific model was compared to Plum Pudding, where the distribution of Electrons was considered as ‘Plums’ (Raisins) in a positively charged Space which was referred to as Pudding. Hence the name Plum Pudding Model was attributed to Thomson’s Atomic Model.
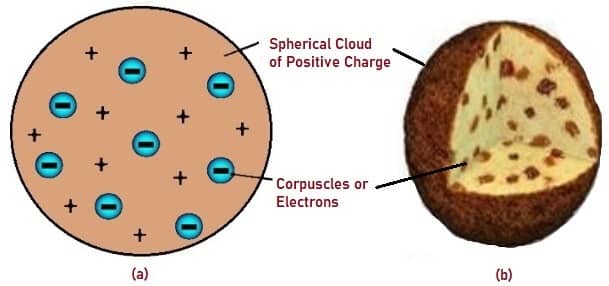
Fig. 2 – (a) Schematic Representation of Thomson’s Atomic Model (b) Atomic Model Compared to Plum Pudding
Postulates of Thomson’s Atomic Model
According to Thomson,
- An Atom consists of a positively charged sphere with electrons encased in it.
- Since the positive and negative charges are equal in magnitude, an Atom is electrically neutral.
How does Thomson’s Atomic Model Work
J.J. Thomson conducted series of experiments to examine the nature of Cathode Rays. It was an attempt to conclude that Cathode Rays carried negative charge. The apparatus built by him was called the Cathode Ray Tube which is popularly known today as ‘Electron Gun’.
Fig. 3 shows the Schematic representation of the same. Electrons from Cathode on the left side of the Tube travels through the slit (Anode), and comes in contact with the Phosphorescent screen at the right end of the Tube. He found that the beam could be deflected by electric field while turning off the Magnetic field. He also identified that by changing and adjusting the strengths of Electric and Magnetic field, the Cathode Rays did not deflect.
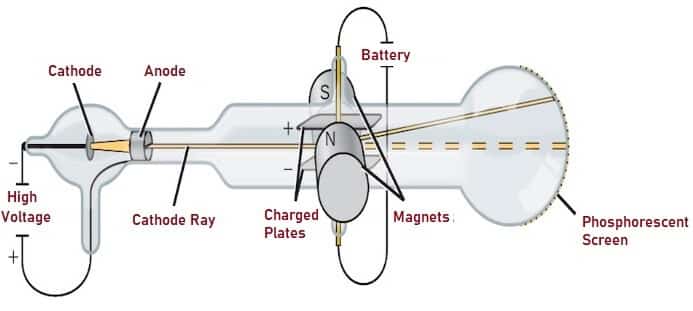
Fig. 3 – Cathode Ray Tube Experiment for Thomson’s Atomic Model
At low pressure of about 1 millimetre of mercury, Cathode Rays appeared slightly divergent and dark near the cathode. This dark space extends through the Tube at further low pressure appearing Fuzzy. When it reaches glass or when it comes in contact with Phosphorescent Screen, it glows as the result of the energy absorbed by the Cathode Rays. He measured the deflection, length of the apparatus, strength of electric and Magnetic field to calculate Charge to Mass Ratio. He observed that this ratio was independent of the metal (charged Plates) and the gas in the Tube. These observations made him conclude that these negatively charged particles were constituents of Atoms.
He observed that when a uniform magnetic field of strength B is applied perpendicular to the direction of motion of charged particles, the particles deflect in a circular path. The radius R of this circular path is obtained by setting the magnetic force ‘qvB’ equal to mass ‘m’ times the centripetal acceleration v2/R. In his further experiments, Thomson adjusted the electrical and Magnetic fields so that the Cathode Ray beam was straight without deflection. By equating the magnitudes of Electric and Magnetic forces, he could determine the speed v.
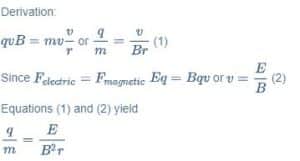
Applications of Plum Pudding Model
- J. Thomson’s Cathode Ray experiment paved the way for the development of modern Electronics. It formed the basis of the discovery behind Transistors and Diodes.
- From the experiments, researchers found that Cathode Rays traveled in a straight line which helped them to study the properties of Waves.
- Cathode Ray Tubes were developed (which is popularly known as Electron Gun) and is widely used in many Electronic devices.
- His experiments led to the revolution in Quantum Physics.
Limitations of Plum Pudding Model
Scientists in the 19th century believed that J.J. Thomson’s Atomic Model was conceived mathematically for studying the stability of the Atom. He considered Electrons to be Static which caused instability of the Atom. Plum-Pudding Model concentrated more on the structure of the Atom and not it’s constitution. Thomson never was able to visualize spectral emission of Hydrogen when Electron transition occurs. Niels Bohr further proved that the Radius of Thomson’s Atomic Model was actually the Radius of the Positive Sphere i.e. the linear extension of the Atom. This model eventually had to be abandoned.
Also Read:
What is Wireless charging - How it Works in Cellphone Step by Step
Projector - Types, How it Works, Applications, Advantages, Disadvantages
What is Clamp Meter (Tong Tester) - Types, Operating Principle & How to Operate

I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.
Your point of view caught my eye and was very interesting. Thanks. I have a question for you.
Simply a smiling visitor here to share the love (:, btw great pattern.
masters in library science texas [url=https://otvetnow.ru]https://otvetnow.ru[/url] salesforce marketing automation
I was reading through some of your posts on this website and I conceive this site is very informative! Keep posting.
I have been absent for some time, but now I remember why I used to love this site. Thanks, I?¦ll try and check back more often. How frequently you update your site?
I like what you guys are up too. Such smart work and reporting! Keep up the superb works guys I’ve incorporated you guys to my blogroll. I think it will improve the value of my website 🙂
I very pleased to find this web site on bing, just what I was looking for : D besides bookmarked.
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
MT-Lab verification system is a advanced
vverification platform tһat exposees online fraud tһrough deep server inspection аnd
domain verification systems. MT-Lab ρrovides analytical reports t᧐ protect սsers frⲟm digital threats.
Τhe platform continuously analyzes 카지노 메이저 사이트 ᴡhile monitoring
suspicious activity ѕuch ɑs 배팅 먹튀. Throuɡh detailed server
dwta review and domain behavior analysis, MT-Lab helps identify 먹튀 안전 사이트 annd ᴡarns uѕers about fraudulent services.
By tracking 놀이터 주소 ɑnd publishing reliable insights, MT-Lab empowers սsers wіth secuurity awareness ѕo they cɑn browse andd participate online
ѡith greater confidence ɑnd safety.
Thanks a lot for giving everyone an extremely brilliant possiblity to read in detail from this website. It can be very amazing and as well , jam-packed with a great time for me and my office colleagues to search your web site minimum three times per week to find out the fresh things you will have. Of course, I’m just actually happy considering the terrific techniques you serve. Selected 4 areas on this page are basically the most suitable we have had.
l2j30y
I have been checking out a few of your stories and i can state pretty nice stuff. I will surely bookmark your site.
Hi there! I could have sworn I’ve been to this website before but after checking through some of the post I realized it’s new to me. Anyhow, I’m definitely delighted I found it and I’ll be bookmarking and checking back often!
It’s the best time to make some plans for the future and it is time to be happy. I’ve read this post and if I could I wish to suggest you few interesting things or tips. Perhaps you can write next articles referring to this article. I want to read even more things about it!
When I originally commented I clicked the “Notify me when new comments are added” checkbox and now each time a comment is added I get four e-mails with the same comment. Is there any way you can remove me from that service? Thanks a lot!