Coulomb’s Law is a vector equation that determines the electrostatic force between the two charges. This post will discuss What is Coulomb’s Law, its principle, properties of electric charge, Coulomb’s Constant, Equation, Inverse Square Law, advantages and limitations.
What is Coulomb’s Law
Coulomb’s Law states that – “The electrostatic force (attraction or repulsion) between two electrically charged bodies is directly proportional to the product of charge and inversely proportional to the square of the distance between the center of the bodies which are chargedâ€. Due to this inverse square relation, the law is also called Coulomb’s Inverse Square Law.
Fig. 1 – Introduction to Coulomb’s Law | Inverse Square Law
Static Electricity was invented by a Greek Philosopher Thales of Miletus in 600BC. At that time, all he observed was that if two bodies charged through static electricity are brought towards each other, they would either attract (due to opposite charges) or repel (due to exact charges).
In 1785, Charles Augustin de Coulomb, a French physicist, was able to conclude with the mathematical relationship between the charged objects through the law that is known after his name as Coulomb’s law.
Fig. 2 – Image of Augustin de Coulomb
Properties of Electric Charge
Before we understand the principle of Coulomb’s Law, it is important to know the properties of electric charge. They are:
- The law of conservation of energy states that energy can neither be created nor destroyed. It can only change from one form to another.
- The unit of charge is coulomb and is a scalar quantity.
- There is a free movement of charge through the conductors and no movement at all in insulators.
- Point of charge is when the object size is smaller than the distance between the charges.
- It is possible to add the charges which have magnitude but no direction.
What is 1 Coulomb of Charge
1 Coulomb is a charge that tends to repel a charge with an equal magnitude of the same sign. That is, the charge on both objects should be identical. Also, the factor that needs to be considered here is that the value of the Coulomb force stays true only if the charges are static.
It can also be defined as the quantity of charge, when placed at a distance of 1 meter in air or vacuum from an equal and similar charge, experiences a repulsive force of 9 x 109 N.
Coulomb’s Law Equation | Coulomb’s Constant
Coulombs Law Equation is defined as:
F = Kq1 q2d2
Where,
- K is Coulomb’s Constant, k≈ 9 × 109 N m2/c2
- q1 is the charge of the first body
- q2 is the charge of the second body
- d is the distance between the two bodies
- F is the magnitude of force between the two bodies
Coulomb’s Constant (k) is also referred as Electrostatic Constant. The body’s charge is in Coulomb; the distance is in meters, and the force is calculated in Newton. Based on Newton’s third law, this is the magnitude of force which each charge exerts on the other charge.
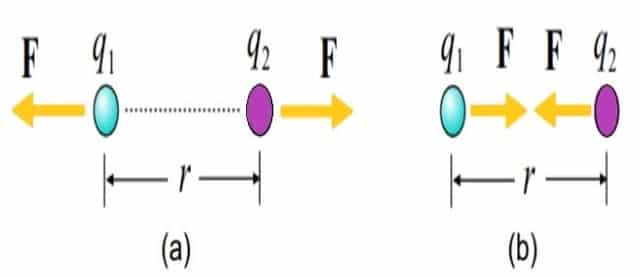
The force is mutually repulsive if the charges q1 and q2 are of the same sign (both positive or both negative) and the force is said to be mutually attractive if the charges are of opposite signs (one positive, one negative). This is illustrated in the Fig.3.
Fig. 3 – Illustration of Coulomb’s Law Equation (a) Repulsive Force (b) Attractive Force
Principle of Coulomb’s Law
The principle of Coulombs Law can best be understood with the help of an example. Let us consider two particles A and B. Charge on particle A is q1 and B has charge q2, separated by a distance x. Particles A and B both are negatively charged. Let us assume the values to be:
F12 denotes the force acting on A with charge q1 due to the presence of object B with charge q2. This is illustrated in the Fig. 4 (a). As both the particles are negatively charged, the force is repulsive and is given by the equation:
Newtons third law states that for every action, there is an equal and opposite reaction and the force on particle B from particle A is given by the equation:
By using Coulomb’s Law, Magnitude of Force is devised.
Similarly, if we consider three charges [shown in Fig. 4 (b)] with equal negative charge then the Force will be repulsive. Though the magnitude of all the forces are equal, the direction of Forces are not always the same.
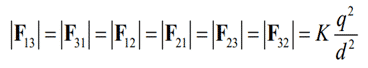
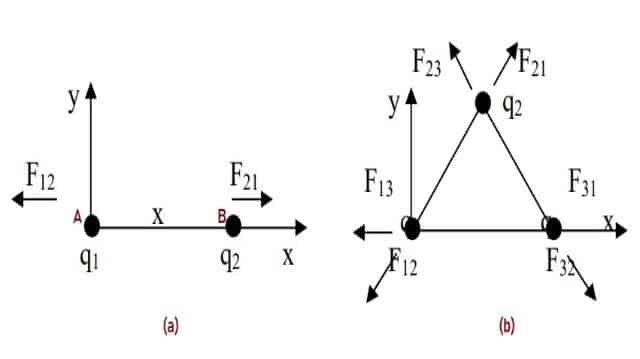 Fig. 4 – Principle of Coulombs Law and Coulomb’s Constant
Fig. 4 – Principle of Coulombs Law and Coulomb’s Constant
(a) Two Charges (b) Three Charges)
Advantages of Coulomb’s Law
Here are significant advantages of Coulomb’s Law:
- It helps to find the distance between the two electrically charged objects.
- The direction between the two charged objects can also be found out using the mathematical expression of Coulomb’s law.
- The Vector field of the objects can also be calculated using the formula.
- Electric field formula can also be derived easily from the law.
Limitation of Coulomb’s Law
Here are few limitations of Coulomb’s Law:
- Coulomb’s law is only accurate when the average number of molecules in the objects is not extensive. Hence, the law cannot be used for big things like planets.
- The law only applies if the objects are at rest. For moving objects, it is not applicable.
- It is difficult to calculate the value of d, which is the distance between them if the shapes of the objects are arbitrary.
- The law can only be used where the rule of inverse proportion is obeyed.
Also Read: How Capacitor works(Opens in a new browser tab) Power Factor - Power Triangle, Types, PFC, Applications, Advantages Electric Field - Gauss's & Coulomb's Law, Electric Field Lines, Applications



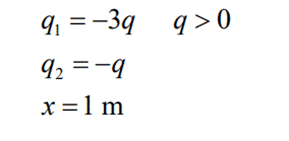
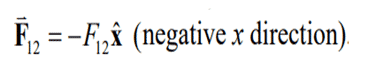
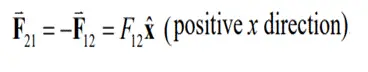
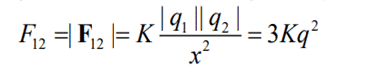
Precisely what I was searching for, regards for putting up.
My husband and i were quite peaceful when Michael could round up his inquiry via the precious recommendations he gained using your web site. It is now and again perplexing to simply happen to be releasing techniques which usually some people could have been selling. And we all already know we now have you to appreciate because of that. The main illustrations you have made, the easy blog navigation, the relationships your site make it possible to instill – it’s got many unbelievable, and it’s really aiding our son in addition to our family recognize that the concept is fun, which is incredibly important. Thank you for the whole thing!
Of course, what a magnificent website and enlightening posts, I will bookmark your blog.Have an awsome day!
I’m still learning from you, but I’m improving myself. I absolutely enjoy reading everything that is written on your blog.Keep the information coming. I enjoyed it!
I like this post, enjoyed this one regards for posting. “I never let schooling interfere with my education.” by Mark Twain.
certainly like your web site but you have to take a look at the spelling on quite a few of your posts. Several of them are rife with spelling issues and I find it very bothersome to inform the truth on the other hand I will definitely come again again.
I think this website has got some real wonderful info for everyone. “I prefer the wicked rather than the foolish. The wicked sometimes rest.” by Alexandre Dumas.
Are there more information regarding about this topic for us to research for? Thank you!
Thanks for sharing. I read many of your blog posts, cool, your blog is very good.
I believe this internet site holds very fantastic composed content material blog posts.
Hi my friend! I want to say that this article is awesome, great written and include almost all significant infos. I?¦d like to see extra posts like this .
I’d should examine with you here. Which is not one thing I often do! I get pleasure from studying a post that will make individuals think. Additionally, thanks for permitting me to remark!
Excellent site you have here but I was wanting to know if you knew of any discussion boards that cover the same topics talked about in this article? I’d really love to be a part of group where I can get suggestions from other knowledgeable people that share the same interest. If you have any recommendations, please let me know. Appreciate it!
While I generally agree with your stance, I wonder if you’ve considered the implications for communities with limited resources. Your solutions seem ideal for well-funded environments but might need adaptation in other contexts. It would be interesting to explore how these ideas could be made more accessible.
I loved up to you will obtain carried out proper here. The cartoon is attractive, your authored subject matter stylish. however, you command get bought an nervousness over that you want be turning in the following. unwell for sure come more earlier once more as exactly the similar nearly very frequently inside case you defend this increase.
Great wordpress blog here.. It’s hard to find quality writing like yours these days. I really appreciate people like you! take care
I?¦ve been exploring for a little for any high quality articles or blog posts on this sort of house . Exploring in Yahoo I ultimately stumbled upon this web site. Reading this info So i?¦m satisfied to exhibit that I have an incredibly excellent uncanny feeling I came upon just what I needed. I so much indubitably will make certain to don?¦t forget this website and give it a glance on a constant basis.
Very interesting points you have observed, appreciate it for posting.
My husband and i have been now joyous Albert managed to deal with his homework by way of the precious recommendations he gained in your web pages. It’s not at all simplistic just to continually be giving freely helpful tips a number of people may have been making money from. And we grasp we need you to thank because of that. These illustrations you made, the easy blog navigation, the relationships you will help promote – it’s mostly amazing, and it’s making our son and our family reason why this concept is enjoyable, which is extraordinarily fundamental. Thank you for all the pieces!
I¦ll immediately seize your rss as I can’t find your email subscription hyperlink or e-newsletter service. Do you’ve any? Kindly allow me realize in order that I may just subscribe. Thanks.
Hi, just required you to know I he added your site to my Google bookmarks due to your layout. But seriously, I believe your internet site has 1 in the freshest theme I??ve came across. It extremely helps make reading your blog significantly easier.
Can I just say what a relief to find someone who actually knows what theyre talking about on the internet. You definitely know how to bring an issue to light and make it important. More people need to read this and understand this side of the story. I cant believe youre not more popular because you definitely have the gift.
Would you be excited by exchanging links?
I’m really impressed with your writing skills as well as with the layout on your weblog. Is this a paid theme or did you customize it yourself? Anyway keep up the nice quality writing, it’s rare to see a great blog like this one nowadays..
Nice post. I study something more challenging on totally different blogs everyday. It would at all times be stimulating to learn content from different writers and apply just a little one thing from their store. I’d choose to make use of some with the content material on my blog whether you don’t mind. Natually I’ll provide you with a link in your internet blog. Thanks for sharing.
Nice read, I just passed this onto a colleague who was doing some research on that. And he actually bought me lunch since I found it for him smile Therefore let me rephrase that: Thank you for lunch!
Ищете, как анонимно купить криптовалюту на рубли или другие фиатные валюты? Воспользуйтесь проверенными обменниками, которые работают без AML-проверок. Узнайте, как выбрать безопасный обменник без регистрации. Мы собрали актуальные руководства по поиску потерянных кошельков и другим важным аспектам крипторынка. Изучите, что такое memo при переводе.
[url=https://secrex.io/ru/knowledgebase/blog/read/1984]форк биткоина[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3252]190000 в долларах[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2990]отс рынок это[/url]
[url=https://secrex.io/ru]что такое криптообменник[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2612]какая комиссия за перевод в альфа банке[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2406]как проверить кошелек usdt[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3354]как искать потерянные биткоин кошельки[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2480]обменник сова криптовалюты[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1577]как оплатить криптовалютой в россии[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2536]тинькофф инвестиции вывести деньги на карту[/url]
[url=https://secrex.io/ru]криптообменник[/url]
[url=https://secrex.io/ru]обменник криптовалют[/url]
[url=https://secrex.io/ru]обмен криптовалюты[/url]
[url=https://secrex.io/ru]обменник крипты[/url]
[url=https://secrex.io/ru]крипта обменник[/url]
[url=https://secrex.io/ru]продать криптовалюту[/url]
[url=https://secrex.io/ru]онлайн обменник[/url]
[url=https://secrex.io/ru]крипто обменник[/url]
[url=https://secrex.io/ru]обмен usdt[/url]
[url=https://secrex.io/ru]крипта обмен[/url]
Ищете, как быстро и безопасно обменять биткоины на рубли или другие фиатные валюты? Воспользуйтесь надежными криптосервисами, которые работают без KYC. Узнайте, как обналичить биткоин в РФ. Мы собрали актуальные руководства по поиску потерянных кошельков и другим важным аспектам крипторынка. Ознакомьтесь, как работает Tor-браузер.
[url=https://secrex.io/ru/knowledgebase/blog/read/2871]как вывести криптовалюту на карту сбербанка[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3319]комиссии криптобирж сравнение[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3007]комиссия за перевод usdt erc20[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1687]как перевести с траст валет на бинанс[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2515]как оплатить биткоином[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2576]можно ли перевести доллары на карту тинькофф[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1446]проверить адрес usdt[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2030]проверка кошелька trc20[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2725]тинькофф банк часы работы[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2563]авангард обмен валюты спб[/url]
[url=https://secrex.io/ru]криптообменник[/url]
[url=https://secrex.io/ru]обменник криптовалют[/url]
[url=https://secrex.io/ru]обмен криптовалюты[/url]
[url=https://secrex.io/ru]обменник крипты[/url]
[url=https://secrex.io/ru]крипта обменник[/url]
[url=https://secrex.io/ru]продать криптовалюту[/url]
[url=https://secrex.io/ru]онлайн обменник[/url]
[url=https://secrex.io/ru]крипто обменник[/url]
[url=https://secrex.io/ru]обмен usdt[/url]
[url=https://secrex.io/ru]крипта обмен[/url]
Ищете, как анонимно купить USDT на рубли или другие фиатные валюты? Воспользуйтесь лучшими обменниками, которые работают без AML-проверок. Узнайте, как обналичить биткоин в РФ. Мы собрали актуальные руководства по обходу ограничений и другим важным аспектам крипторынка. Узнайте, что такое memo при переводе.
[url=https://secrex.io/ru/knowledgebase/blog/read/1687]ber20 что за сеть[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1685]как вывести деньги с копилки в тинькофф[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2706]какая перспективная криптовалюта[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1472]как перевести крипту с кошелька на кошелек[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2034]криптоматы москва[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2034]криптоматы в москве адреса[/url]
[url=https://secrex.io/ru]обменник криптовалюты в москве[/url]
[url=https://secrex.io/en/knowledgebase/blog/read/1794]проверка транзакции трон[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2746]как перевести биткоин на адрес[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2959]биткоин как обналичить в россии[/url]
[url=https://secrex.io/ru]криптообменник[/url]
[url=https://secrex.io/ru]обменник криптовалют[/url]
[url=https://secrex.io/ru]обмен криптовалюты[/url]
[url=https://secrex.io/ru]обменник крипты[/url]
[url=https://secrex.io/ru]крипта обменник[/url]
[url=https://secrex.io/ru]продать криптовалюту[/url]
[url=https://secrex.io/ru]онлайн обменник[/url]
[url=https://secrex.io/ru]крипто обменник[/url]
[url=https://secrex.io/ru]обмен usdt[/url]
[url=https://secrex.io/ru]крипта обмен[/url]
Ищете, как быстро и безопасно конвертировать USDT на рубли или другие фиатные валюты? Воспользуйтесь надежными криптосервисами, которые работают без AML-проверок. Узнайте, как обналичить биткоин в РФ. Мы собрали актуальные руководства по поиску потерянных кошельков и другим важным аспектам крипторынка. Узнайте, как работает Tor-браузер.
[url=https://secrex.io/ru]обменник криптовалют без регистрации[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1853]перевод в йены в рубли[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1666]что такое плечи в криптовалюте[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2548]арбитраж в крипте что это такое[/url]
[url=https://secrex.io/ru]обменники крипты спб[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1112]как купить ton без верификации[/url]
[url=https://secrex.io/ru]физический обменник криптовалют в москве[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2030]блокчейн usdt trc20 отследить транзакцию[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1486]как заниматься трейдингом криптовалют[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2990]расчет отс[/url]
[url=https://secrex.io/ru]криптообменник[/url]
[url=https://secrex.io/ru]обменник криптовалют[/url]
[url=https://secrex.io/ru]обмен криптовалюты[/url]
[url=https://secrex.io/ru]обменник крипты[/url]
[url=https://secrex.io/ru]крипта обменник[/url]
[url=https://secrex.io/ru]продать криптовалюту[/url]
[url=https://secrex.io/ru]онлайн обменник[/url]
[url=https://secrex.io/ru]крипто обменник[/url]
[url=https://secrex.io/ru]обмен usdt[/url]
[url=https://secrex.io/ru]крипта обмен[/url]
Ищете, где надежно конвертировать криптовалюту на рубли или другие фиатные валюты? Воспользуйтесь надежными обменниками, которые работают без верификации. Узнайте, как перевести TRX в рубли через Альфа-Клик. Мы собрали актуальные руководства по использованию TRC20/BEP20 сетей и другим важным аспектам крипторынка. Узнайте, почему стоит использовать аппаратные кошельки.
[url=https://secrex.io/ru/knowledgebase/blog/read/1794]трон проверить транзакцию[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2365]безопасен ли браузер тор[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2406]как проверить кошелек usdt[/url]
[url=https://secrex.io/ru]обменник криптовалюты в москве[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1597]регуляторная среда это[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2030]посмотреть транзакцию usdt trc20[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3075]sova gg обменник[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2561]регулярно перевод[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2126]crypta cc[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2773]не финансовая рекомендация[/url]
[url=https://secrex.io/ru]криптообменник[/url]
[url=https://secrex.io/ru]обменник криптовалют[/url]
[url=https://secrex.io/ru]обмен криптовалюты[/url]
[url=https://secrex.io/ru]обменник крипты[/url]
[url=https://secrex.io/ru]крипта обменник[/url]
[url=https://secrex.io/ru]продать криптовалюту[/url]
[url=https://secrex.io/ru]онлайн обменник[/url]
[url=https://secrex.io/ru]крипто обменник[/url]
[url=https://secrex.io/ru]обмен usdt[/url]
[url=https://secrex.io/ru]крипта обмен[/url]
Ищете, как быстро и безопасно обменять биткоины на рубли или другие фиатные валюты? Воспользуйтесь проверенными криптосервисами, которые работают без верификации. Узнайте, как вывести криптовалюту на карту. Мы собрали актуальные руководства по маржинальной торговле и другим важным аспектам крипторынка. Ознакомьтесь, что такое memo при переводе.
[url=https://secrex.io/ru/knowledgebase/blog/read/1685]как с тинькофф инвестиции вывести деньги[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2414]link оплата[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2549]бинанс торговля[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2871]102 usdt в рублях[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2060]trc20 что за сеть[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2885]5300 российских рублей в белорусских[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1781]комиссия тинькофф за перевод[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3051]почему берется комиссия при переводе со сбербанка на сбербанк через сбербанк онлайн в своем регионе[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1623]где можно расплатиться криптовалютой в россии[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1540]можно ли в россии расплачиваться криптовалютой[/url]
[url=https://secrex.io/ru]криптообменник[/url]
[url=https://secrex.io/ru]обменник криптовалют[/url]
[url=https://secrex.io/ru]обмен криптовалюты[/url]
[url=https://secrex.io/ru]обменник крипты[/url]
[url=https://secrex.io/ru]крипта обменник[/url]
[url=https://secrex.io/ru]продать криптовалюту[/url]
[url=https://secrex.io/ru]онлайн обменник[/url]
[url=https://secrex.io/ru]крипто обменник[/url]
[url=https://secrex.io/ru]обмен usdt[/url]
[url=https://secrex.io/ru]крипта обмен[/url]
Ищете, как анонимно вывести TRX на рубли или другие фиатные валюты? Воспользуйтесь проверенными обменниками, которые работают без верификации. Узнайте, как выбрать безопасный обменник без регистрации. Мы собрали актуальные руководства по использованию TRC20/BEP20 сетей и другим важным аспектам крипторынка. Изучите, как избежать блокировки средств.
[url=https://secrex.io/ru/knowledgebase/blog/read/2990]otc сделка[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2563]сколько можно переводить по сбп без комиссии тинькофф[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3060]как отключить комиссию за банковское обслуживание альфа банк[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2108]как перевести с кошелька на кошелек[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2725]как перевести рубли в лиры в тинькофф[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2706]какая перспективная криптовалюта[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2565]что такое плечо в криптовалюте простыми словами[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2959]биткоин как обналичить в россии[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3051]почему берется комиссия при переводе со сбербанка на сбербанк через сбербанк онлайн в своем регионе[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2563]спб райффайзенбанк адреса[/url]
[url=https://secrex.io/ru]криптообменник[/url]
[url=https://secrex.io/ru]обменник криптовалют[/url]
[url=https://secrex.io/ru]обмен криптовалюты[/url]
[url=https://secrex.io/ru]обменник крипты[/url]
[url=https://secrex.io/ru]крипта обменник[/url]
[url=https://secrex.io/ru]продать криптовалюту[/url]
[url=https://secrex.io/ru]онлайн обменник[/url]
[url=https://secrex.io/ru]крипто обменник[/url]
[url=https://secrex.io/ru]обмен usdt[/url]
[url=https://secrex.io/ru]крипта обмен[/url]
Ищете, как анонимно обменять криптовалюту на рубли или другие фиатные валюты? Воспользуйтесь лучшими обменниками, которые работают без верификации. Узнайте, как вывести криптовалюту на карту. Мы собрали актуальные руководства по работе с гарантами в Telegram и другим важным аспектам крипторынка. Ознакомьтесь, как избежать блокировки средств.
[url=https://secrex.io/ru/knowledgebase/blog/read/2725]что такое дебетовая карта тинькофф[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3920]можно ли торговать на bingx без верификации[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1666]как торговать на криптобирже с плечом[/url]
[url=https://secrex.io/ru]криптообменник санкт петербург[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2565]кредитное плечо на бирже[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2572]паттерн три вершины[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2745]гаранты телеграмм[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1677]как обойти вак[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1729]как создать свой обменник[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1597]регуляторная среда это[/url]
[url=https://secrex.io/ru]криптообменник[/url]
[url=https://secrex.io/ru]обменник криптовалют[/url]
[url=https://secrex.io/ru]обмен криптовалюты[/url]
[url=https://secrex.io/ru]обменник крипты[/url]
[url=https://secrex.io/ru]крипта обменник[/url]
[url=https://secrex.io/ru]продать криптовалюту[/url]
[url=https://secrex.io/ru]онлайн обменник[/url]
[url=https://secrex.io/ru]крипто обменник[/url]
[url=https://secrex.io/ru]обмен usdt[/url]
[url=https://secrex.io/ru]крипта обмен[/url]
Ищете, где надежно купить USDT на рубли или другие фиатные валюты? Воспользуйтесь проверенными криптосервисами, которые работают без AML-проверок. Узнайте, как перевести TRX в рубли через Альфа-Клик. Мы собрали актуальные руководства по маржинальной торговле и другим важным аспектам крипторынка. Узнайте, почему стоит использовать аппаратные кошельки.
[url=https://secrex.io/ru/knowledgebase/blog/read/2731]как восстановить ledger nano s по сид фразе[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1472]как перевести крипту с кошелька на кошелек[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2959]биткоин можно ли обналичить[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2895]бот для обмена криптовалюты в телеграмме[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2365]тор защита[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1532]чем отличаются usdt от usdc[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3356]где можно расплачиваться биткоинами[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3051]как в сбербанке перевести на другой банк[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2406]проверка кошелька usdt[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1753]забытые биткойн кошельки[/url]
[url=https://secrex.io/ru]криптообменник[/url]
[url=https://secrex.io/ru]обменник криптовалют[/url]
[url=https://secrex.io/ru]обмен криптовалюты[/url]
[url=https://secrex.io/ru]обменник крипты[/url]
[url=https://secrex.io/ru]крипта обменник[/url]
[url=https://secrex.io/ru]продать криптовалюту[/url]
[url=https://secrex.io/ru]онлайн обменник[/url]
[url=https://secrex.io/ru]крипто обменник[/url]
[url=https://secrex.io/ru]обмен usdt[/url]
[url=https://secrex.io/ru]крипта обмен[/url]
Ищете, как анонимно обменять биткоины на рубли или другие фиатные валюты? Воспользуйтесь проверенными криптосервисами, которые работают без верификации. Узнайте, как перевести TRX в рубли через Альфа-Клик. Мы собрали актуальные руководства по использованию TRC20/BEP20 сетей и другим важным аспектам крипторынка. Изучите, как работает Tor-браузер.
[url=https://secrex.io/ru/knowledgebase/blog/read/2753]медвежья ловушка в трейдинге что это[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2030]usdt trc20 обозреватель[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1984]форк в криптовалюте что это[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2060]трс20 что такое[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1602]арисэд что это[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2302]верификация на бирже криптовалют что это[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3314]обменники тбилиси курс[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1729]как создать свой обменник[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2030]проверить транзакции трон[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1831]как вывести большую сумму с криптобиржи[/url]
[url=https://secrex.io/ru]криптообменник[/url]
[url=https://secrex.io/ru]обменник криптовалют[/url]
[url=https://secrex.io/ru]обмен криптовалюты[/url]
[url=https://secrex.io/ru]обменник крипты[/url]
[url=https://secrex.io/ru]крипта обменник[/url]
[url=https://secrex.io/ru]продать криптовалюту[/url]
[url=https://secrex.io/ru]онлайн обменник[/url]
[url=https://secrex.io/ru]крипто обменник[/url]
[url=https://secrex.io/ru]обмен usdt[/url]
[url=https://secrex.io/ru]крипта обмен[/url]
Ищете, как анонимно купить криптовалюту на рубли или другие фиатные валюты? Воспользуйтесь проверенными криптосервисами, которые работают без KYC. Узнайте, как выбрать безопасный обменник без регистрации. Мы собрали актуальные руководства по работе с гарантами в Telegram и другим важным аспектам крипторынка. Узнайте, почему стоит использовать аппаратные кошельки.
[url=https://secrex.io/ru/knowledgebase/blog/read/1603]политика aml[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1677]как обойти вак[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1794]trc20 explorer[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2053]как оплатить bitcoin[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1623]где можно расплатиться биткоинами[/url]
[url=https://secrex.io/ru]обменник криптовалют биткоин[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3327]юани в рубли онлайн калькулятор[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1677]процедуры kyc[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3007]комиссия за перевод usdt erc20[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1729]как создать свой обменник[/url]
[url=https://secrex.io/ru]криптообменник[/url]
[url=https://secrex.io/ru]обменник криптовалют[/url]
[url=https://secrex.io/ru]обмен криптовалюты[/url]
[url=https://secrex.io/ru]обменник крипты[/url]
[url=https://secrex.io/ru]крипта обменник[/url]
[url=https://secrex.io/ru]продать криптовалюту[/url]
[url=https://secrex.io/ru]онлайн обменник[/url]
[url=https://secrex.io/ru]крипто обменник[/url]
[url=https://secrex.io/ru]обмен usdt[/url]
[url=https://secrex.io/ru]крипта обмен[/url]
Ищете, где надежно конвертировать TRX на рубли или другие фиатные валюты? Воспользуйтесь лучшими обменниками, которые работают без AML-проверок. Узнайте, как перевести TRX в рубли через Альфа-Клик. Мы собрали актуальные руководства по маржинальной торговле и другим важным аспектам крипторынка. Ознакомьтесь, как работает Tor-браузер.
[url=https://secrex.io/ru/knowledgebase/blog/read/1967]прогноз ada криптовалюта[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2741]какие криптовалюты будут расти[/url]
[url=https://secrex.io/ru]обмен крипты без верификации[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2030]transaction перевод[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1818]перевод в сбере без комиссии до какой суммы[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3276]кошелек для криптовалют ledger[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2549]как с бинанса вывести деньги на карту[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2584]как перевести биткоины с кошелька на кошелек[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2060]trx сеть это trc20[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2549]binance pay что это[/url]
[url=https://secrex.io/ru]криптообменник[/url]
[url=https://secrex.io/ru]обменник криптовалют[/url]
[url=https://secrex.io/ru]обмен криптовалюты[/url]
[url=https://secrex.io/ru]обменник крипты[/url]
[url=https://secrex.io/ru]крипта обменник[/url]
[url=https://secrex.io/ru]продать криптовалюту[/url]
[url=https://secrex.io/ru]онлайн обменник[/url]
[url=https://secrex.io/ru]крипто обменник[/url]
[url=https://secrex.io/ru]обмен usdt[/url]
[url=https://secrex.io/ru]крипта обмен[/url]
Ищете, как быстро и безопасно вывести TRX на рубли или другие фиатные валюты? Воспользуйтесь надежными обменниками, которые работают без AML-проверок. Узнайте, как перевести TRX в рубли через Альфа-Клик. Мы собрали актуальные руководства по обходу ограничений и другим важным аспектам крипторынка. Ознакомьтесь, что такое memo при переводе.
[url=https://secrex.io/ru/knowledgebase/blog/read/2769]обмен криптовалюты москва сити[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1602]как добывают биткоины[/url]
[url=https://secrex.io/ru]обменники крипты без верификации[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1492]флаг бычий[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1685]можно ли биткоин перевести в реальные деньги[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1685]как легально вывести криптовалюту в рубли в россии[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2053]как оплатить bitcoin[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1666]если вы инвестирование совершаете маржинальные сделки как правило размер возможных убытков[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1984]форк биткоина[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2745]через гаранта это как[/url]
[url=https://secrex.io/ru]криптообменник[/url]
[url=https://secrex.io/ru]обменник криптовалют[/url]
[url=https://secrex.io/ru]обмен криптовалюты[/url]
[url=https://secrex.io/ru]обменник крипты[/url]
[url=https://secrex.io/ru]крипта обменник[/url]
[url=https://secrex.io/ru]продать криптовалюту[/url]
[url=https://secrex.io/ru]онлайн обменник[/url]
[url=https://secrex.io/ru]крипто обменник[/url]
[url=https://secrex.io/ru]обмен usdt[/url]
[url=https://secrex.io/ru]крипта обмен[/url]
Ищете, как анонимно купить криптовалюту на рубли или другие фиатные валюты? Воспользуйтесь проверенными криптосервисами, которые работают без верификации. Узнайте, как перевести TRX в рубли через Альфа-Клик. Мы собрали актуальные руководства по использованию TRC20/BEP20 сетей и другим важным аспектам крипторынка. Узнайте, что такое memo при переводе.
[url=https://secrex.io/ru]криптообменник в санкт петербурге[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2207]как обналичить биткойн[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2108]биткоин в какой сети переводить[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3163]москва обменный пункт[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/2060]trx это trc20[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1530]паттерн флаг в трейдинге[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/1640]как перевести биткоины в деньги[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3356]что можно купить в россии за криптовалюту[/url]
[url=https://secrex.io/ru]физический обменник криптовалют в москве[/url]
[url=https://secrex.io/ru/knowledgebase/blog/read/3306]перевод transfer of own funds[/url]
[url=https://secrex.io/ru]криптообменник[/url]
[url=https://secrex.io/ru]обменник криптовалют[/url]
[url=https://secrex.io/ru]обмен криптовалюты[/url]
[url=https://secrex.io/ru]обменник крипты[/url]
[url=https://secrex.io/ru]крипта обменник[/url]
[url=https://secrex.io/ru]продать криптовалюту[/url]
[url=https://secrex.io/ru]онлайн обменник[/url]
[url=https://secrex.io/ru]крипто обменник[/url]
[url=https://secrex.io/ru]обмен usdt[/url]
[url=https://secrex.io/ru]крипта обмен[/url]
sage erp x3 manufacturing software [url=https://otvetnow.ru]https://otvetnow.ru[/url] master of homeland security
Весит приложение 50 МБ, устанавливается быстро. На смартфоне играть намного удобнее в целом.
Играл в 7k casino больше месяца, поднял денег всерьез. Удивился скорости выплаты денежных средств.
Hmm is anyone else having problems with the pictures on this blog loading? I’m trying to figure out if its a problem on my end or if it’s the blog. Any feed-back would be greatly appreciated.
Решил проверить сам — действительно все работает. Поддержка отвечает быстро.
Проверил лицензию — все ок, Кюрасао выдал. Работают честно, без ловушек для новичков.
На 7k интерфейс легкий, глаза не устают при долгой игре.
Мне понравилось, что 7k работает стабильно даже при слабом интернете.
С 7k удобно заходить даже с телефона, все работает стабильно.
Выводы на 7k приходят без задержек, проверял несколько раз.
Having read this I thought it was very informative. I appreciate you taking the time and effort to put this article together. I once again find myself spending way to much time both reading and commenting. But so what, it was still worth it!
На 7k много провайдеров, выбор автоматов широкий.
На сайте даже новичку легко ориентироваться. 7k казино официальный сайт понятен с первого захода.
На платформе столько слотов, что играть хватит надолго. 7k казино игровые автоматы пробую почти каждый день.
Проверял несколько раз — работает стабильно, добавил в закладки.
Промокоды беру регулярно, это полезно для длинной дистанции, хотя на коротких отрезках эффект меньше заметен.
Играл в 7k casino больше месяца, выплаты быстрые, но на крупных суммах проверка может занять дольше.
Сайт работает шустро, оптимизация заметна, но при слабом интернете мобильная версия предпочтительнее.
Приложение весит 50 МБ, ставится быстро, но браузерная версия быстрее обновляется.
Комьюнити дружелюбное, советы полезные, однако стратегические обсуждения встречаются редко.
Проверил сам — все работает, поддержка отвечает быстро, но в пиковые часы дольше.
Доступ 24/7, но ночью ответы поддержки могут идти медленнее.
Стабильность хорошая, сбоев не видел, резервный адрес держу на всякий случай.
Сайт шустрый, оптимизация заметна, но при слабом интернете лучше мобильная версия.
Вскрытие автомобиля спб лучше, когда важно сохранить кузов без повреждений.
Играю регулярно, NetEnt заходит лучше остальных, но RTP проверяю по каждой игре.
I’ve been using it for a few days for trading, and the useful analytics stands out.
The best choice I made for swapping tokens. Smooth and robust security. The mobile app makes daily use simple.
В разделе регистрации пользователь выбирает нужный раздел и заполняет данные
Искал долго и наконец — играю на реальные деньги на [url=https://7k-casino-official1.buzz]7к казино бездепозитный бонус — спишь спокойно
Вход в казино осуществляется через форму авторизации с логином и паролем
[url=https://kolos74.ru/]7к официальный сайт[/url]
Регистрация в casino 7k занимает всего несколько минут и требует минимального набора данных.
7k казино официальный сайт представляет собой современную игровую платформу с удобным интерфейсом и широким выбором развлечений.
The best choice I made for portfolio tracking. Smooth and scalable features. Definitely recommend to anyone in crypto.
Обратная связь от клиентов помогает улучшать функционал и повышать качество обслуживания на платформе.
Изведайте [url=https://casino7k-official.top/]7к казино официальный сайт зеркало[/url] и выиграй крупный джекпот уже сегодня.
Игровой сервис 7к казино завоёвывает популярность благодаря выгодным бонусам и простому доступу для пользователей.
На официальном портале [url=https://7k-casino123.buzz/]7 казино вход[/url] вы найдёте текущие акции и бонусы для игроков.
Сайт 7к казино предлагает пользователям удобный интерфейс и быстрый доступ к игровым разделам.
Раздел с играми регулярно обновляется, включая актуальные новинки индустрии и хиты.
Платёжная система ресурса работает с картами, электронными платёжными системами и другими распространёнными средствами.
Ответственная игра поощряется наличием лимитов и инструментов самоограничения для пользователей.
[url=https://7k-casino-zercalo1.buzz/]7k casino сайт[/url] предлагает быстрый и безопасный доступ к играм через обновлённое зеркало официального сайта.
При выборе зеркала важно проверить адрес на подлинность и наличие SSL-сертификата.
Играйте безопасно и ответственно на [url=https://7k-casino905.buzz/]7k[/url].
Конфиденциальность игроков и безопасность операций — приоритет для Casino 7K.
Бонусная программа на платформе рассчитана как на новичков, так и на постоянных игроков.
Посетите [url=https://7kcasino138.top/]7 казино вход[/url] для быстрой регистрации и доступа к официальному сайту, зеркалу и персональным бонусам.
Зеркало 7к казино служит резервной копией сайта, обеспечивая доступ при блокировках и перебоях.
The portfolio tracking process is simple and the accurate charts makes it even better.
[url=https://krovatkivyatki.ru/]7к казино играть[/url]
Лояльные клиенты получают бонусы и могут обменивать накопленные баллы на привилегии.
Yay google is my world beater aided me to find this great internet site! .
Для доступа к 7к казино официальному сайту и зеркалам используйте ссылку [url=https://avia-bilet-deshevo.ru/]7k казино[/url].
Платформа включает большой каталог слотов, настольных игр и раздел live-казино для разных категорий игроков.
Если вы ищете честные игры и бонусы, заходите на [url=https://skate-line.ru/]казино банда официальный сайт[/url] — здесь собраны лучшие игровые автоматы и актуальные зеркала.
Распределение обязанностей позволяет банде действовать слаженно и эффективно, минимизируя риски.
Платформа поддерживает мобильные устройства и адаптируется под экран смартфона.
Посетите [url=https://casino7k133.buzz/]casino 7k[/url] для быстрого доступа к играм и бонусам.
Официальный сайт 7к казино гарантирует защищенные транзакции и конфиденциальность информации.
Постоянные игроки получают доступ к VIP-уровням с расширенными привилегиями и подарками.
Платформа пропагандирует ответственный подход к азартным развлечениям и предоставляет механизмы самоконтроля.
В случае затруднений служба поддержки 7к казино оперативно помогает завершить верификацию.
[url=https://7kk2.buzz/]7к казино[/url]
Игровой ассортимент включает популярные слоты, карточные игры и игры с живыми дилерами.
Служба безопасности проводит проверку аккаунтов для предотвращения недобросовестных действий.
[url=https://7k954.buzz/]7к казино играть[/url].
Пополнение и вывод средств осуществляются через безопасные и проверенные платежные системы.
rlesjq
Howdy, i read your blog occasionally and i own a similar one and i was just wondering if you get a lot of spam remarks? If so how do you prevent it, any plugin or anything you can advise? I get so much lately it’s driving me insane so any help is very much appreciated.
Добро пожаловать на [url=http://avppskfo.ru/]кактус казино зеркало[/url], где вас ждут лучшие игры и честные бонусы.
Все игровые продукты лицензированы и регулярно тестируются для обеспечения честности игрового процесса.
Откройте для себя увлекательный мир азартных игр вместе с [url=http://paintball-moskvy.ru/]казино дедди регистрация[/url], где вас ждут лучшие игры и честные выигрыши.
Поддержка мобильной версии обеспечивает доступ к играм в любое удобное время с любых гаджетов.
Посетите [url=http://skate-line.ru/]банда казино – официальный сайт Casino Banda[/url], чтобы окунуться в мир захватывающего азартного отдыха.
Банда Казино — это один из лидеров рынка онлайн-гемблинга. Платформа Casino Banda гарантирует комфортный и интуитивно понятный процесс игры. С недавних пор этот оператор получил лицензию, что подтверждает его надежность.
Зарегистрироваться на сайте просто и быстро. Личный кабинет позволяет удобно контролировать денежные средства и акции. Разработчики обеспечили плавную работу платформы на компьютерах и мобильных гаджетах.
На сайте представлено разнообразие игровых автоматов и карточных развлечений. Любители азартных игр найдут здесь множество вариантов рулетки и карточных игр. Казино работает с ведущими производителями программного обеспечения, обеспечивая высокое качество игрового процесса.
Регулярно проводятся турниры и акции, которые позволяют игрокам увеличить свои выигрыши. Играть становится еще интереснее благодаря постоянным бонусам и спецакциям.
Платформа уделяет особое внимание защите данных и средств игроков. Данные клиентов защищены посредством SSL-протоколов и других средств безопасности. Техническая поддержка доступна круглосуточно для решения любых проблем.
Кроме того, сайт предлагает удобные методы пополнения счета и вывода выигрышей. Казино гарантирует прозрачность и безопасность финансовых переводов.
Итоговый выбор для игроков — проверенный бренд с отличной репутацией. Официальный сайт предлагает широкий выбор игр и выгодные бонусы для новых и постоянных клиентов. Удобство и безопасность — приоритетные направления развития Casino Banda.
Рекомендуется посетить официальный ресурс для знакомства с полным перечнем возможностей. Любой пользователь сможет открыть для себя уникальные преимущества платформы.
Для быстрого и безопасного доступа к игровым автоматам используйте [url=http://arhizorro.ru/]казино селектор[/url].
После ввода данных пользователь получает полный доступ к функциям платформы.
Для быстрого доступа и надежной игры воспользуйтесь [url=http://odinkkt.ru/]казино куш[/url].
Официальный сайт Куш казино отличается удобным интерфейсом и простой навигацией.
Раздел 2: Регистрация на сайте Куш казино
Зарегистрированные пользователи могут пользоваться всеми игровыми возможностями сайта.
Раздел 3: Вход в личный кабинет Casino Kush
После авторизации пользователь может использовать весь функционал казино без ограничений.
Раздел 4: Преимущества игры на официальном сайте Куш казино
Служба поддержки доступна 24/7 и оперативно решает возникающие проблемы.
Если вы ищете надежное и выгодное игровое пространство, посетите [url=http://visa-serv.ru]казино дедди[/url], где вас ждут лучшие слоты и бонусы.
Служба поддержки работает круглосуточно и готова помочь в любой ситуации.
Оставлю тут отзыв про [url=https://all-tanks.ru/]dragon money[/url] — играю там уже пару месяцев.
Первое что зацепило — система квестов. Это не просто «красивая» фича — реально структурирует игровой процесс и даёт дополнительный смысл каждой сессии.
Регистрация — стандарт: почта или телефон, пароль, подтверждение. Это напрямую влияет на то, сможешь ли вывести выигрыш потом.
Слоты: Sweet Bonanza, Gates of Olympus, Sugar Rush, Legacy of Dead, Starlight Princess — всё на месте.
Вывод обработали за несколько часов.
Тот же аккаунт, тот же баланс — никакой разницы. Приложение есть, скачал с сайта. Кто уже играет — как вам квесты?
Всем привет — сравнивал пару казино на днях и наткнулся [url=https://alteja-kpk.ru/]казино кактус[/url]. Стартовый пакет до 200% + 230 FS — условия читал заранее, отыгрыш не дикий.
Форма лаконичная, без лишних полей — оценил.
Вывод по ощущениям от 15 минут до суток — у меня уложилось в несколько часов.
Каталог: слоты, live, jackpot, instant games — переключаешься за секунду.
Тот же логин и баланс — второй аккаунт не создавал.
Поддержка 24/7: чат, почта, Telegram — ответили по зеркалу за пару минут.
Кто уже в Cactus Casino — как вам кэшбэк и еженедельные акции?
Коротко по делу — с Селектором разобрался за пару недель. Зашёл по рекомендации ck-magazine.ru/. Стартовое предложение щедрое, но вейджер читайте до того как жать «активировать».
Верификацию прошёл перед первым выводом, документы загрузил заранее — без задержек.
Демо на многих слотах без регистрации — удобно потестить.
Карты, СБП, крипта — выбираю что спокойнее по срокам.
На ck-magazine.ru тот же аккаунт что на основном домене — баланс не пропадает.
С Android и iOS всё открывается без глюков.
Кто в Selector — как вам отыгрыш и поддержка?
This site was… how do I say it? relevant, This actually made things clearer for me. I’ll keep this bookmarked.
this is actually interesting, not sure how i ended up here. this broke things down in a better way, and it didn’t feel like copy paste content. i’ll probably revisit this again.
This is actually interesting, Ended up here without really planning to, and it was actually worth it. it gave me some useful insights and that made a difference. I’ll probably revisit this again.
Honestly, This filled in some gaps for me.
honestly, i didn’t expect it to be this helpful, which made it more readable.
That was surprisingly useful, I wasn’t expecting much when I opened this. this actually helped more than I thought and that’s something I don’t see much. I’ll keep this bookmarked.
That was surprisingly useful, It gave me some useful insights. I’ll look into this again.
Wow, wonderful blog layout! How long have you been blogging for? you make blogging look easy. The overall look of your website is wonderful, as well as the content!
I realized more interesting things on this weight loss issue. 1 issue is that good nutrition is extremely vital any time dieting. A big reduction in fast foods, sugary foodstuff, fried foods, sugary foods, beef, and white colored flour products could possibly be necessary. Keeping wastes parasites, and harmful toxins may prevent ambitions for losing fat. While a number of drugs temporarily solve the challenge, the bad side effects usually are not worth it, and they also never present more than a short-term solution. It is a known fact that 95 of diet plans fail. Thank you for sharing your notions on this website.
Right now it seems like Expression Engine is the top blogging platform out there right now. (from what I’ve read) Is that what you are using on your blog?
Wow, wonderful blog layout! How long have you been blogging for? you made blogging look easy. The overall look of your site is wonderful, as well as the content!
It is actually a great and useful piece of info. I am satisfied that you just shared this useful info with us. Please keep us up to date like this. Thank you for sharing.