Rutherford’s Atomic Model also called as ‘Nuclear Model’ was devised by Ernest Rutherford who was an English physicist and an accomplished experimentalist. This post will discuss in detail about what is Rutherford’s Atomic Model (Nuclear Model), Gold Foil experiment, its results, applications and limitations.
What is Rutherford’s Atomic Model
In Rutherford’s time, the Plum Pudding Model of an Atom was widely accepted. They believed that Atom’s positive charge and mass were spread out evenly. However, many scientists around the world proved this theory to be wrong.
In 1911, Rutherford, Geiger and Marsden performed the Gold Foil experiment, also called Geiger-Marsden experiment which was a breakthrough in the field of chemistry.
Fig. 1 – Introduction to Rutherford’s Atomic Model
He came up with a different Hypotheses and he stated that Atom has mostly empty space and the positive charge is concentrated in a dense mass at its core, forming a Nucleus. He is known to the world as the “Father of Nuclear Physicsâ€. This collaborative effort of all the scientists was put forward based on the scattering theory of α-particles which later came to be known as Rutherford’s Atomic Model also called as Nuclear Model.
Rutherford also discovered Radioactivity when he was experimenting with two thin sheets of metal in 1899. There he discovered two distinct elements α- radiation and β-radiation. Shortly after, he detected a third type of radiation called as γ-radiation.
Fig. 2 – Ernest Rutherford
Rutherford’s Gold Foil Experiment (Geiger-Marsden Experiment)
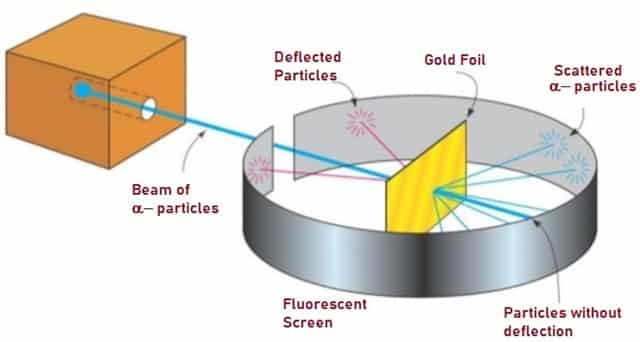
Rutherford teamed up with his assistant, Hans Geiger and Ernst Marsden who was an undergraduate student working in Rutherford’s lab; conducted “Gold Foil Experiment†also known as the Geiger-Marsden experiment. His idea was to probe the structure of Atom by firing α-particles or helium ions, at thin metal foils (Gold Foil).
Rutherford expected that the fraction of particles scattered through an angle of θ or greater should have been observed to fall off like a Gaussian; if Plum Pudding Atomic Model was correct.
In this experiment, Rutherford used Gold Foil which was extremely thin sheet, not more than 1000 atoms thick. Gold Foil was bombarded with positively charged α-particles.
He used α-particles because they are very energetic and are heavier than other particles. They are approximately 4 times heavier than Hydrogen atoms. Lead container acted as source for α-particles. Since he thought that the atom had a pudding-like structure, with electrons and positively charged particles embedded in it, he expected that, the α-particles would just pass through.
Fig. 3 – Gold Foil Experiment
Instead he found that the α-particles scattered through different angles. The scattered particles were detected by the fluorescent screen which emitted fluorescent spikes. He observed that most of the particles passed through the gold foil with very little deflection or no deflection at all.
Few particles scattered at large angles i.e. the particles traveled in the opposite direction. This experiment proved that the entire mass of an Atom is located at the center and was named Nucleus. Hence ‘Rutherford’s Atomic Model’ is also called as ‘Nuclear Model’.
Results and Conclusion of Gold Foil Experiment
Rutherford came out with a different hypothesis after the experiment and the results ruled out the Plum Pudding structure of an Atom. The conclusions for this hypothesis were:
- An Atom is mainly empty inside. This came from the fact that there were a number of α-particles that traveled in a straight line.
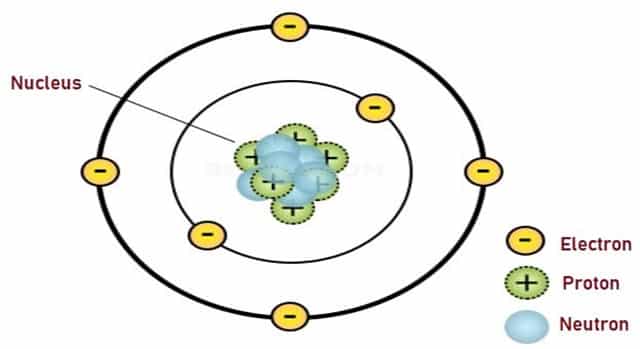
- There has to be a central Nucleus in an Atom which is positively charged. Nearly all the mass of an Atom is found in the Nucleus. He concluded this because of those α-particles that got deflected once they reached the atoms of the gold foil. α-particles are positive, so is the Nucleus; positives repel.
- The Electrons revolve around the Nucleus in a circular motion.
Based on this, Rutherford’s Atomic Model came into existence. He concluded that all the mass of the Atom and all the positive particles embedded in it are all contained in this infinitely tiny mass which he called the Nucleus. Nucleus is a word that comes from the Latin word “nucula†meaning “little nutâ€.
Fig. 4 – Rutherford’s Atomic Model
These conclusions, however, were also later confirmed as inaccurate. We know that an object travelling in constant circular motion gains acceleration. If according to Rutherford’s hypothesis the electrons gained acceleration in an Atom they would have released some form of energy which would have finally taken over the Nucleus and caused great instability inside the Atom.
Rutherford’s hypothesis needed some slight modifications since we know that in nature all Atoms are stable. Later on, Danish physicist Neils Bohr proposed his theory which proved that the Atoms are stable. Rutherford’s studies and conclusions have led us today to learn more about the Atom, which can be defined as the smallest particle of a chemical element in existence.
Applications of Rutherford’s Atomic Model
The applications include:
- Rutherford’s crucial discovery of Nucleus marked the birth of Nuclear Physics.
- Experiments conducted by Rutherford paved the way for Nuclear Fission experiments which were used for war and peace.
- Nuclear Fission principle is used in Nuclear Reactors.
- The radioactive particles as used and discovered by Rutherford α-alpha, β-beta and γ-gamma particles led to major breakthrough in Healthcare industry.
- Gradually, Radioactive sources were implemented to supervise industrial processes like cement industries, Food and Agriculture industry.
- Rutherford discovered the principle of half-life period for Radioactive elements.
- Based on Rutherford’s theory, Radium-226, Strontium-90, Cobalt-60 which are the sources of α, β, γ particles respectively are produced commercially.
Limitations of Rutherford’s Atomic Model
The limitations include:
- This model failed as the Atoms produced discrete line spectrum instead of continuous spectrum.
- According to this model, Atoms are unstable which is false.
Also Read:
What is ATOM - Atomic Structure, Atomic Models and Applications
Reliability fundamentals
Thomson’s Atomic Model – How it Works, Postulates and Limitations



Your point of view caught my eye and was very interesting. Thanks. I have a question for you.
fao47p
I’ve been exploring for a little for any high-quality articles or weblog posts in this kind of area . Exploring in Yahoo I eventually stumbled upon this site. Reading this information So i am glad to exhibit that I’ve an incredibly excellent uncanny feeling I came upon exactly what I needed. I most undoubtedly will make sure to do not omit this website and give it a glance on a relentless basis.
of course like your web site but you have to check the spelling on quite a few of your posts. Many of them are rife with spelling issues and I in finding it very bothersome to tell the reality then again I¦ll surely come again again.
Woh I enjoy your content, saved to favorites! .
We are a group of volunteers and starting a brand new scheme in our community. Your website provided us with helpful info to work on. You have performed a formidable process and our entire group will be thankful to you.
a-authentic garage door [url=https://otvetnow.ru]https://otvetnow.ru[/url] blinncollege
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
Magnificent web site. A lot of useful information here. I am sending it to a few buddies ans also sharing in delicious. And of course, thank you to your sweat!
Howdy! I’m at work surfing around your blog from my new iphone 3gs! Just wanted to say I love reading through your blog and look forward to all your posts! Carry on the superb work!
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me?
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me?
Hi would you mind letting me know which web host you’re using? I’ve loaded your blog in 3 different web browsers and I must say this blog loads a lot faster then most. Can you recommend a good web hosting provider at a honest price? Thanks a lot, I appreciate it!
It’s a shame you don’t have a donate button! I’d most certainly donate to this brilliant blog! I suppose for now i’ll settle for book-marking and adding your RSS feed to my Google account. I look forward to new updates and will share this website with my Facebook group. Chat soon!
I’ve been exploring for a little bit for any high-quality articles or blog posts on this sort of area . Exploring in Yahoo I at last stumbled upon this website. Reading this info So i am happy to convey that I’ve an incredibly good uncanny feeling I discovered just what I needed. I most certainly will make certain to do not forget this website and give it a look on a constant basis.
Definitely believe that that you stated. Your favourite justification appeared to be on the internet the easiest factor to understand of. I say to you, I certainly get annoyed at the same time as people consider issues that they just don’t realize about. You managed to hit the nail upon the top and outlined out the entire thing with no need side effect , folks could take a signal. Will probably be again to get more. Thank you
4ep70j
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
Good info. Lucky me I reach on your website by accident, I bookmarked it.
Yeah bookmaking this wasn’t a risky conclusion great post! .
My spouse and I stumbled over here coming from a different web address and thought I might as well check things out. I like what I see so i am just following you. Look forward to going over your web page for a second time.