Electrons that occupy the outermost orbit of an Atom are called Valence Electrons. This post will discuss in detail about Valence Electrons, Electron Dot Diagrams, Characteristics, Energy Bands.
What are Valence Electrons
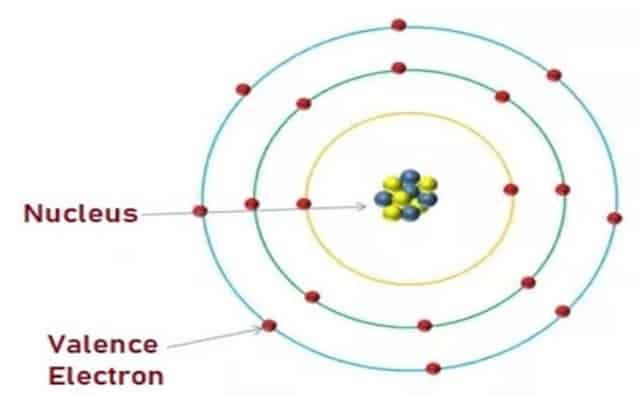
Valence Electrons occupy the orbit that is farthest from the Nucleus of an Atom and this orbit is called a Valence Shell. These Electrons involve or participate in forming compounds’ ionic bonds or chemical bonds. The properties of the atom are determined by the number of Electrons in the valence shell.
Richard Abegg was a pioneer in Valence theory and in the year 1904, he proposed Abegg’s Rule which stated that, the difference of the maximum positive and negative valences of an element is usually eight. This theory was later used by Gilbert Lewis and he came up with “Octet Ruleâ€. Octet Rule states that an Atom is more stable when it has 8 Electrons. Octet can be achieved by its own Electrons or by sharing. Atoms continue to form bonds until it reaches Noble gas electron configuration i.e., 8 Electrons in the outermost shell.
Fig. 2 – (a) Gilbert Lewis (b) Richard AbeggÂ
Electron Dot Diagrams
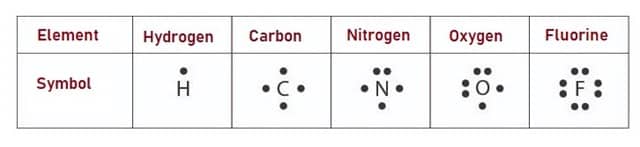
The electrons in the outermost shell of an Atom can be represented by using dots around the symbol of an element. This representation is called as Lewis Electron Dot Diagram. Number of dots indicates number of atoms of an element. Any element can have maximum of 8 Electrons and hence the number of dots also should be 8. Fig. 2 shows Electron Dot Diagrams.
Fig. 3 – Electronic Dot Diagram of Different Elements
Characteristics of Valence Electrons
The characteristics are:
- An Atom which has less than 4 Valence Electrons (VE), tend to lose/give up electrons to form positive Cations.
- An Atom which has more than 4 VE, tends to gain/borrow electrons to form negative Anions.
- For atoms with exactly 4 VE, it can go either way.
- For any Atom with 8 VE, is considered as the most stable form.
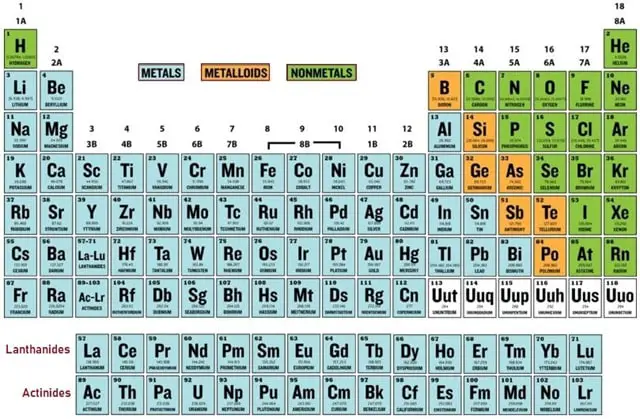
Fig. 4 – Periodic Table
Determining VE using Periodic Table
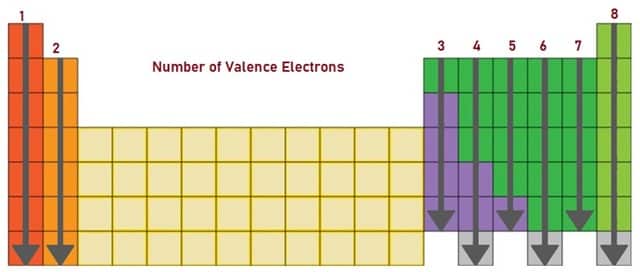
The position of the Elements in the Periodic Table reflects the number of Valence-Electrons. Fig. 4 shows Periodic Table in which the elements within the same group or in the same column will have the same number of Valence-Electrons and they also have similar chemical properties. If you observe the Periodic Table carefully, you can see that the number of Valence Electrons in groups 1 and 2 and 13 to 18 increases by one from one element to the next. The number is represented in Fig. 5.
How do Valence Electrons Conduct Electricity
Since these Electrons are far from the Nucleus, they are loosely held in the orbit. And also, they require minimal force to leave the Atom and occupy the Valence-shell of the other Atom. This “flow†of “Electrons†is known as Electrical Current. It is measured in Amperes and the force required by the Electron to exit from its Valence Shell is called as EMF (Electro-Motive Force) and is measured in Volts.
Fig. 6 – Electrons Conduct Electricity
Energy Band
Energy Bands can be defined as the group of individual energy levels of electrons surrounding each atom. The concept of energy bandgap can be understood better by knowing two different energy levels namely:
- Valence Band
- Conduction Band
Valence Band
This band consists of Valence Electrons present in the Valence Shell of an Atom. It requires minimal energy to free itself from the outer shell and become free electrons. These Free Electrons move further to Conduction Band.
Conduction Band
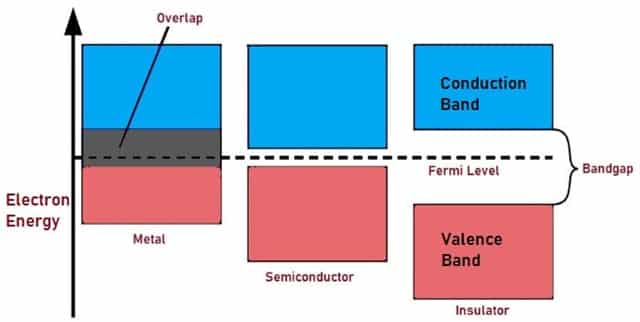
The free electrons that are responsible for conduction are present in Conduction Band. The Electrons that leave the valence Band reach higher energy levels or conduction band in order to help in Conduction. This band is above Fermi level. Fermi level or Fermi energy is defined as the highest energy level that the electron occupies at zero temperature.
Fig. 7 – Representation of Energy Band
Band Gap or energy gap is different for conductors, insulators and semiconductors as shown in the Fig. 7. Band gap is very less for conductors and hence the Electrons move easily to conduction band from Valence Band. For semi-conductors, the energy gap exists between the two bands and more energy is required to free Valence electrons. For Insulators, there exists large gap between Conduction Band and Valence Band.
Valence electrons are the highest energy electrons in an atom whereas Core Electrons are tightly bound and are unaffected by chemical reactions as they are closer to the Nucleus. Hope you found this article worth reading!
Also Read: Bohr's Atomic Model - Postulates of Neil Bohr Atomic Model & Limitations Rutherford's Atomic Model - Gold Foil Experiment, Results & Applications Flywheel as Energy Storage Device, Calculations and Rotor Requirements



9brad6
oubv7l
37mbsp
mz7cfl
I think this is among the most vital information for me. And i am glad reading your article. But want to remark on few general things, The site style is ideal, the articles is really nice : D. Good job, cheers
v7p6ba
ut36p2
hiq7vv
1mbs94
0micw0
v38cpx
a62wm6
I am happy that I noticed this site, exactly the right info that I was looking for! .
6m1ci9
I am always invstigating online for articles that can assist me. Thanks!
You are my intake, I have few blogs and rarely run out from brand :). “Fiat justitia et pereat mundus.Let justice be done, though the world perish.” by Ferdinand I.
Would you be curious about exchanging hyperlinks?
Having read this I thought it was very informative. I appreciate you taking the time and effort to put this article together. I once again find myself spending way to much time both reading and commenting. But so what, it was still worth it!
Just wanna remark that you have a very nice site, I love the style it really stands out.
hul204
What¦s Going down i am new to this, I stumbled upon this I have found It absolutely helpful and it has helped me out loads. I hope to contribute & aid other customers like its helped me. Good job.
ns55mj
b0geze
j2sysg
You should take part in a contest for one of the best blogs on the web. I will recommend this site!
Heya i am for the first time here. I came across this board and I find It really useful & it helped me out a lot. I hope to give something back and aid others like you helped me.
Very efficiently written article. It will be supportive to everyone who utilizes it, including me. Keep up the good work – for sure i will check out more posts.
8ixmvi
m4g1ae
Excellent post however I was wondering if you could write a litte more on this topic? I’d be very grateful if you could elaborate a little bit more. Kudos!
x46yca
Merely wanna remark on few general things, The website design and style is perfect, the subject matter is really great. “Earn but don’t burn.” by B. J. Gupta.
You made some good points there. I did a search on the subject and found most people will approve with your blog.
Thank you for another informative blog. Where else could I get that kind of information written in such a perfect way? I have a project that I’m just now working on, and I have been on the look out for such info.
Some truly superb info , Gladiolus I discovered this. “What you do speaks so loudly that I cannot hear what you say.” by Ralph Waldo Emerson.
Great ?V I should certainly pronounce, impressed with your site. I had no trouble navigating through all the tabs and related info ended up being truly simple to do to access. I recently found what I hoped for before you know it at all. Quite unusual. Is likely to appreciate it for those who add forums or anything, site theme . a tones way for your customer to communicate. Nice task..
whoah this blog is magnificent i love reading your articles. Keep up the great work! You know, a lot of people are looking around for this information, you could aid them greatly.
You are my inhalation, I own few web logs and very sporadically run out from to brand : (.
You have brought up a very good points, thanks for the post.
Thanks for sharing
I do like the manner in which you have presented this particular issue plus it really does provide me some fodder for thought. Nonetheless, coming from what I have observed, I basically wish as other feedback stack on that people today continue to be on issue and don’t embark on a tirade of the news du jour. Still, thank you for this fantastic point and whilst I can not really concur with this in totality, I value the perspective.
Only wanna comment on few general things, The website design and style is perfect, the subject material is really excellent : D.
Definitely, what a fantastic blog and enlightening posts, I definitely will bookmark your site.Have an awsome day!
I love how you backed up every point with real examplesmade the whole topic so relatable and easy to understand. Thanks for putting in the time to create this!
An impressive share, I simply given this onto a colleague who was doing a little analysis on this. And he actually purchased me breakfast as a result of I discovered it for him.. smile. So let me reword that: Thnx for the deal with! But yeah Thnkx for spending the time to discuss this, I really feel strongly about it and love reading more on this topic. If possible, as you become experience, would you mind updating your blog with extra details? It’s highly useful for me. Huge thumb up for this blog publish!
I as well think thence, perfectly pent post! .
Howdy! Quick question that’s entirely off topic. Do you know how to make your site mobile friendly? My blog looks weird when viewing from my apple iphone. I’m trying to find a theme or plugin that might be able to resolve this problem. If you have any suggestions, please share. Cheers!
Hi, i think that i saw you visited my weblog thus i came to “return the favor”.I am trying to find things to improve my site!I suppose its ok to use a few of your ideas!!
You are a very intelligent individual!
Volleys, spectacular strikes and their conversion rates
This is a topic close to my heart cheers, where are your contact details though?
I like this web site so much, saved to bookmarks.
Good write-up, I’m normal visitor of one’s site, maintain up the excellent operate, and It’s going to be a regular visitor for a long time.
There’s certainly a lot to find out about this topic. I like all of the points you made.
一饭封神在线免费在线观看,海外华人专属平台采用机器学习个性化推荐,高清无广告体验。
I feel that is one of the so much important information for me. And i’m glad reading your article. But want to observation on few basic issues, The site taste is perfect, the articles is in reality nice : D. Good process, cheers
Great article, totally what I wanted to find.
Have you ever considered creating an e-book or guest authoring on other blogs? I have a blog centered on the same information you discuss and would love to have you share some stories/information. I know my audience would appreciate your work. If you are even remotely interested, feel free to shoot me an email.
This is a topic that is close to my heart… Many thanks! Exactly where are your contact details though?
Thank you for the sensible critique. Me & my neighbor were just preparing to do a little research on this. We got a grab a book from our area library but I think I learned more from this post. I’m very glad to see such excellent info being shared freely out there.
pktspy
海外华人必备的iyifan平台结合大数据AI分析,提供最新高清电影、电视剧,无广告观看体验。
If you wish for to obtain much from this post then you have to apply such methods to your won blog.
Hello, yes this paragraph is actually nice and I have learned lot of things
from it concerning blogging. thanks.
pd2oaz
Nice answers in return of this matter with real arguments and describing everything regarding that.
Thanks for a marvelous posting! I definitely enjoyed reading it, you may be a great author.I will make certain to bookmark your blog and will often come back sometime soon. I want to encourage yourself to continue your great posts, have a nice evening!
Heya i am for the first time here. I found this board and I find It truly useful & it helped me out a lot. I hope to give something back and aid others like you helped me.
Unquestionably believe that which you said. Your favorite justification appeared to be at the internet the simplest factor to consider of. I say to you, I certainly get irked while other folks think about issues that they plainly don’t realize about. You controlled to hit the nail upon the highest as well as defined out the whole thing with no need side-effects , other people could take a signal. Will probably be back to get more. Thanks
Hello, yeah this article is actually fastidious and I have learned lot of things from it concerning blogging. thanks.
What’s up, of course this post is really nice and I have learned lot of things from it on the topic of blogging. thanks.
Hi there, just wanted to tell you, I enjoyed this blog post. It was practical. Keep on posting!
This article gives a clear introductory overview of Valence Electron and explains well how these outer electrons determine chemical bonding and many atomic properties. The references to the work of Richard Abegg and Gilbert N. Lewis, especially the connection to the Octet Rule, help place the concept in its historical and theoretical context.
One particularly useful part is the transition from basic chemistry to solid-state ideas such as the Valence Band and Conduction Band. This connection helps readers understand why valence electrons are also crucial for electrical conductivity in materials like semiconductors.
That said, some explanations are simplified. For example, the statement that atoms with fewer than four valence electrons “tend to lose electrons” is a useful rule of thumb but does not apply universally. Overall, however, the article works well as a beginner-friendly explanation of a key concept in chemistry and electronics.
oznjcp
Hi my family member! I wish to say that this post is awesome, nice written and include approximately all vital infos. I’d like to look more posts like this .
I always look forward to your posts, they never disappoint.
Thanks for taking the time to write such a detailed and helpful post.
Remarkable things here. I am very satisfied to see your post. Thank you a lot and I’m looking forward to contact you. Will you kindly drop me a mail?
Amazing blog! Do you have any tips and hints for aspiring writers? I’m hoping to start my own website soon but I’m a little lost on everything. Would you recommend starting with a free platform like WordPress or go for a paid option? There are so many options out there that I’m completely confused .. Any tips? Thanks a lot!
Hi there! Someone in my Facebook group shared this website with us so I came to take a look. I’m definitely loving the information. I’m bookmarking and will be tweeting this to my followers! Fantastic blog and excellent design.
Right now it seems like WordPress is the top blogging platform available right now. (from what I’ve read) Is that what you are using on your blog?
o8a7x2
What’s up to every single one, it’s genuinely a fastidious for me to pay a visit this web page, it includes valuable Information.
Hi there! Quick question that’s completely off topic. Do you know how to make your site mobile friendly? My website looks weird when browsing from my iphone4. I’m trying to find a template or plugin that might be able to resolve this issue. If you have any suggestions, please share. Thanks!
iavfow
wonderful points altogether, you simply won a new reader. What could you suggest in regards to your submit that you just made some days in the past? Any certain?
Hi there, just wanted to tell you, I loved this article. It was helpful. Keep on posting!
Usually I do not read post on blogs, however I wish to say that this write-up very compelled me to check out and do it! Your writing taste has been surprised me. Thanks, quite great post.
After study a few of the blog posts on your website now, and I truly like your way of blogging. I bookmarked it to my bookmark website list and will be checking back soon. Pls check out my web site as well and let me know what you think.
Wow! Thank you! I continuously wanted to write on my site something like that. Can I implement a part of your post to my website?
Your style is very unique compared to other people I’ve read stuff from. Many thanks for posting when you’ve got the opportunity, Guess I will just bookmark this blog.
Hello! Do you use Twitter? I’d like to follow you if that would be okay. I’m absolutely enjoying your blog and look forward to new updates.
Hello There. I discovered your blog the usage of msn. This is an extremely smartly written article. I’ll be sure to bookmark it and come back to read extra of your helpful information. Thank you for the post. I will certainly comeback.
First off I would like to say excellent blog! I had a quick question which I’d like to ask if you don’t mind. I was interested to know how you center yourself and clear your head prior to writing. I’ve had a hard time clearing my thoughts in getting my thoughts out. I truly do take pleasure in writing but it just seems like the first 10 to 15 minutes tend to be lost just trying to figure out how to begin. Any suggestions or tips? Appreciate it!
I like the valuable information you provide in your articles. I will bookmark your blog and check again here frequently. I am quite certain I’ll learn plenty of new stuff right here! Good luck for the next!
You actually make it seem so easy with your presentation but I find this topic to be actually something that I think I would never understand. It seems too complex and extremely broad for me. I am looking forward for your next post, I’ll try to get the hang of it!
I’m not that much of a online reader to be honest but your sites really nice, keep it up! I’ll go ahead and bookmark your site to come back later. All the best
Howdy just wanted to give you a quick heads up. The words in your article seem to be running off the screen in Safari. I’m not sure if this is a formatting issue or something to do with browser compatibility but I thought I’d post to let you know. The layout look great though! Hope you get the issue solved soon. Cheers
Generally I do not read post on blogs, but I wish to say that this write-up very compelled me to check out and do so! Your writing taste has been surprised me. Thanks, quite nice post.
This post will assist the internet visitors for setting up new blog or even a blog from start to end.
Thank you, I have just been looking for information about this topic for ages and yours is the best I have came upon so far. However, what in regards to the bottom line? Are you positive about the supply?
If you desire to grow your know-how only keep visiting this site and be updated with the most up-to-date news posted here.
Thanks for sharing superb informations. Your site is so cool. I’m impressed by the details that you have on this website. It reveals how nicely you perceive this subject. Bookmarked this web page, will come back for extra articles. You, my friend, ROCK! I found just the info I already searched everywhere and just could not come across. What a great site.
hi!,I like your writing very much! share we communicate more about your article on AOL? I require an expert on this area to solve my problem. May be that’s you! Looking forward to see you.
I was wondering if you ever thought of changing the structure of your website? Its very well written; I love what youve got to say. But maybe you could a little more in the way of content so people could connect with it better. Youve got an awful lot of text for only having one or 2 images. Maybe you could space it out better?
Hello just wanted to give you a brief heads up and let you know a few of the images aren’t loading correctly. I’m not sure why but I think its a linking issue. I’ve tried it in two different web browsers and both show the same results.
Right now it looks like BlogEngine is the top blogging platform out there right now. (from what I’ve read) Is that what you’re using on your blog?
This website was… how do you say it? Relevant!! Finally I have found something which helped me. Thank you!
hi!,I really like your writing very a lot! percentage we be in contact more approximately your post on AOL? I require an expert in this area to resolve my problem. May be that is you! Looking forward to look you.
g7n2tr