Voltaic Cell is primarily used to produce Electrical energy through chemical reactions. It is also called Galvanic Cell. This post will discuss in detail about what is Voltaic Cell or Galvanic Cell, it’s construction and types, how it works, applications and advantages.
What is Voltaic Cell or Galvanic Cell
Voltaic Cell (Galvanic Cell) is an electrochemical cell which derives electrical energy from spontaneous redox reactions occurring in the cell. The energy is harnessed by separating the reactants from direct contact with each other while ensuring a channel for the Electrons to flow through them.
Fig. 1 – Introduction to Voltaic Cell
The Voltaic Cell was named after its inventor, the famous Italian physicist, Alessandro Volta, in 1799. Based on Luigi Galvani’s phenomenon of Electrochemical change, he invented “The Voltaic Pile” that is classic Voltaic Cell, which was the first Electrical Battery that could consistently supply an electric current to a circuit. Earlier these cells were used to study the Thermodynamic properties of fused salts.
Fig. 2 – (a) Voltaic Pile (b) Alessandro Volta
Construction of Voltaic Cell (Galvanic Cell)
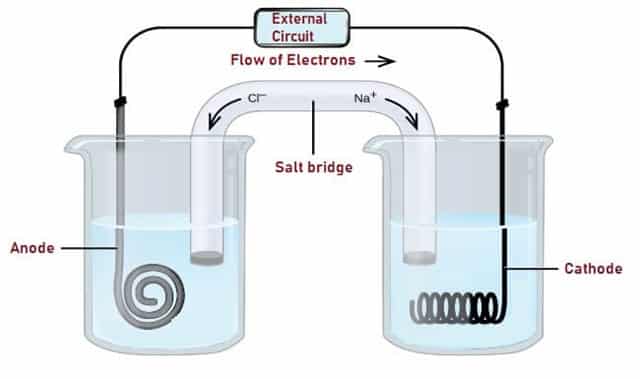
The complete Voltaic Cell consists of two half-cells, normally joined by a semi-permeable membrane or a salt bridge which prevents the ions of the more noble metal from plating out at the other electrode. The electrodes are first immersed in electrolyte solutions.
Then they are connected through an electrical load. It is an Electrochemical Cell with two conductive electrodes. An electrode is a solid electric conductor which supplies electric current into non-metallic solids, liquids or gases etc.
Fig. 3 – Construction of Voltaic Cell
The Voltaic Cell uses two separate metal electrodes each in a solution of electrolytes. The two types of electrodes are anode and cathode. The anode is described as the electrode where oxidation takes place and the cathode is defined as the electrode where reduction occurs. The electrolyte solution contains cations (+) of the electrode metal and anions (−) for balancing cation charges.
Half-cells are nothing but separate slots in which reduction and oxidation takes place. External circuit provides a pathway for the flow of Electrons as shown in the Fig. 3. If both the Electrodes are immersed in the same Electrolyte, then Salt bridge is not required.
Types of Voltaic Cell
They are basically of two types. They are:
- Primary Cell
- Secondary Cell
Primary Cell
A cell which acts as a source of electricity without being previously charged by an electrical current from an external source is called a Primary Cell. In this type of cell, electrical energy is obtained solely by the chemical reactivity as long as the active materials are present. The types of Primary Cell are:
- Dry Cell
- Mercury Cell
- Alkaline cell
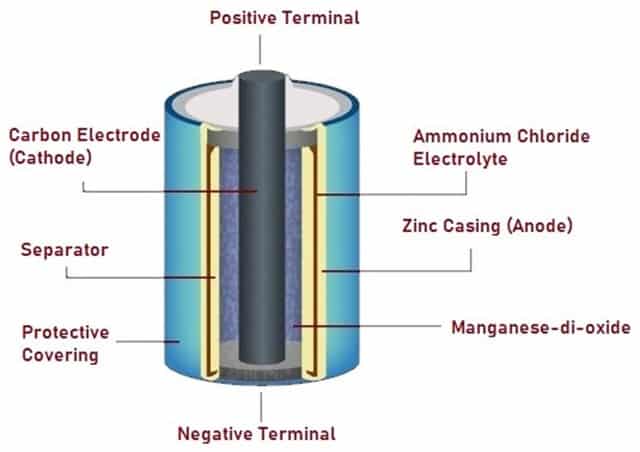
Dry Cell
A Dry Cell is a type of electric battery commonly used for electrical portable devices. It was invented by the German scientist Carl Gassner in 1886 after the invention of wet zinc-carbon batteries by the French engineer Georges Leclanché in 1866. A standard Dry Cell is made up of a zinc anode, which is usually a cylindrical pot, and a carbon cathode in the shape of a central rod.
Fig. 4 – Schematic Diagram of Dry Cell
The electrolyte is Ammonium Chloride next to the Zinc anode, in the form of a paste. Another composition of Ammonium Chloride and Manganese dioxide takes up the remaining space between the electrolyte and the carbon cathode, the latter serving as a depolarizer. The voltage of a Dry Cell battery is 1.5 V.
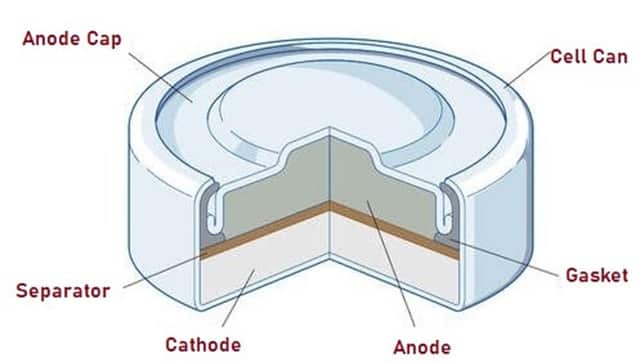
Mercury Cell
Mercury Cells cause a reaction between Mercuric Oxide and Zinc electrodes in an alkaline electrolyte to produce electricity. Mercury Cells are used as button cells for watches, hearing aids, cameras and calculators.
Fig. 5 -Â Schematic Diagram of Mercury Cell
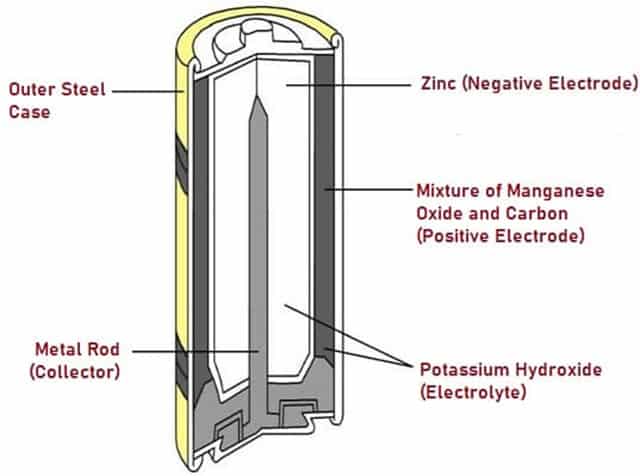
Alkaline Cell
The Alkaline Cells receive their energy by causing a reaction between Zinc metal and Magnesium dioxide. Alkaline cells are named as such because they have an alkaline electrolyte of potassium hydroxide, unlike acidic electrolytes used in many other cells.
Alkaline Cells have the same voltage as dry cells, but have a higher energy density and are more long-lasting than dry cells. Alkaline cells are used MP3 or CD players, cameras, flashlights and toys.
Fig. 6 – Schematic Diagram of Alkaline Cell
Secondary Cell
Secondary Voltaic Cell (Electrical Battery) once used can be recharged by passing a current through it and it may, therefore, be used over and over again. Chemical changes take place when the cell is charged with electricity. These changes are reversed during discharge.
Secondary batteries are classified based on the material used to construct them. They are:
- Nickel-Cadmium Cell
- Lead-Acid Cell
- Lithium-Ion Cell
Fig. 7 – Types of Secondary Cells (a) Nickel-Cadmium Cell (b) Lead-Acid Cell (c) Lithium-Ion Cell
Nickel-Cadmium Cell
Nickel Oxide Hydroxide and Metallic Cadmium electrodes are used in this type of cells. Hence, it is also called NiCd or NiCad batteries.
Lead-Acid Cell
The Lead-Acid type of Voltaic cell is generally used in automobiles. When the cell is fully charged, the negative plate contains lead while the positive plate contains lead dioxide. The electrolyte solution, which is basically a higher concentration of aqueous sulphuric acid, stores most of the chemical energy.
While discharging, both the positive and the negative plates are converted to lead sulphate. The electrolyte loses most of its dissolved sulphuric acid and converts into water.
Lithium-Ion Cell
The Lithium-Ion cells are used in mobile phones. During discharging, the Lithium-Ions move from the negative electrode through an electrolyte to the positive electrode, and vice versa while charging.
How does Voltaic Cell Work
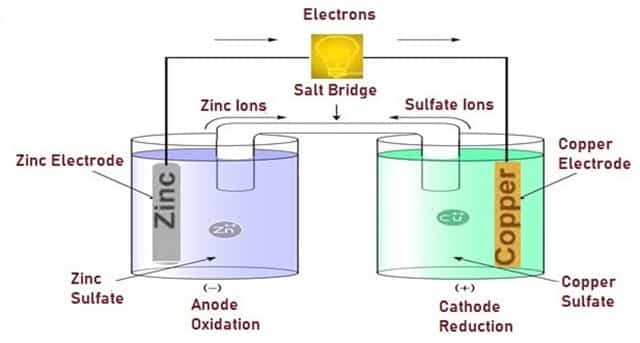
Usually, in a Voltaic Cell, the anode is made with Zinc (Zn) and the cathode is made with Copper. The anode is immersed in a solution of Zinc Sulfate and the cathode is immersed in a solution of Copper Sulfate. To complete the electric circuit a salt bridge is required here.
If the Copper and Zinc electrodes are connected by an external electrical conductor, Zinc from the zinc electrode dissolves into the solution as Zn2+ ions (oxidation), releasing electrons which enter the external conductor.
Fig. 8 – Working of Voltaic Cell
To catch up with the increased Zinc ion concentration, via the Salt bridge, Zinc ions leave and anions enter the Zinc half-cell. Inside the Copper half-cell, the Copper ions plate on the Copper electrode (reduction), take up electrons that leave the external conductor. The electrochemical reaction is:
Zn + Cu2+ → Zn2+ + Cu
At the time of the reaction, the Zinc electrode will be used and the metal will shrink in size, but the Copper electrode will become larger due to the deposited Cu that is being produced. To keep the charge flowing through the cell a salt bridge is required.
Without the salt bridge, the electrons produced at the anode will be deposited at the cathode and the reaction would stop. The electromotive force of the Voltaic cell is the difference of the half-cell potentials, a measure of the relative ease of dissolution of the two electrodes into the electrolyte.
Applications of Voltaic Cell
The applications include:
- They are used in watches, clocks, remote controllers, calculators etc.
- They are also used in cell phones, cameras, laptops.
- They are used as Fuel cells for engines to power up.
- They are also used in toys.
Advantages of Voltaic Cell
The advantages are:
- The secondary cells produce a large and steady amount of electricity for a long time.
- Secondary cells are rechargeable.
- The primary cells are lightweight and hence, they are portable.
- Primary cells provide a steady current and voltage.
Disadvantages of Voltaic Cell
The disadvantages are:
- Secondary cells are expensive.
- The Lead-Acid cells are heavy and acid may spill out.
- Primary cells are not long-lasting.
- Primary cells cannot be recharged.
Also Read: What is Supercapacitor (Ultracapacitor) - Characteristics, Working, Types & Applications Transducer - Characteristics, Type, Application, Factors Influencing Choice Thermistor – Classification, How it Works, Applications and Advantages




Can I just say what a aid to search out someone who actually is aware of what theyre talking about on the internet. You positively know the best way to carry a problem to gentle and make it important. More individuals must learn this and perceive this aspect of the story. I cant believe youre not more well-liked since you definitely have the gift.
iu4wel
Very well explained
Excellent analysis
I believe this website has got some really fantastic information for everyone :D. “A friend might well be reckoned the masterpiece of nature.” by Ralph Waldo Emerson.
Big chances missed, wasteful finishing and conversion failures
I went over this web site and I think you have a lot of excellent information, saved to fav (:.
Thank you, I’ve recently been looking for information about this topic
for ages and yours is the greatest I have found out so far.
But, what about the bottom line? Are you sure concerning the supply?
Highly descriptive blog, I enjoyed that bit. Will there be
a part 2?
Hello there! Do you know if they make any plugins to safeguard against hackers?
I’m kinda paranoid about losing everything I’ve worked hard on. Any recommendations?
Awesome article.
plumber buffalo [url=https://otvetnow.ru]https://otvetnow.ru[/url] new york executive suites
Dead written content, thanks for selective information.
naturally like your web site however you have to take a look at the spelling on several of your posts. Many of them are rife with spelling problems and I in finding it very bothersome to tell the truth then again I’ll surely come again again.
It’s amazing in support of me to have a web site, which is useful for my experience. thanks admin
Usually I don’t read post on blogs, but I wish to say that this write-up very pressured me to try and do so! Your writing style has been amazed me. Thank you, very nice post.
If some one needs to be updated with most recent technologies then he must be go to see this web page and be up to date all the time.
Hello there! This is my first visit to your blog! We are a collection of volunteers and starting a new initiative in a community in the same niche. Your blog provided us valuable information to work on. You have done a wonderful job!
Wow! This could be one particular of the most useful blogs We’ve ever arrive across on this subject. Actually Excellent. I’m also a specialist in this topic so I can understand your effort.
I used to be very pleased to seek out this net-site.I wished to thanks to your time for this excellent learn!! I undoubtedly enjoying every little bit of it and I have you bookmarked to check out new stuff you blog post.
Thanks for one’s marvelous posting! I really enjoyed reading it, you can be a great author.I will make sure to bookmark your blog and will come back sometime soon. I want to encourage that you continue your great work, have a nice evening!
If you are going for finest contents like myself, only visit this website daily since it gives quality contents, thanks
Keep this going please, great job!
Hey! I’m at work surfing around your blog from my new apple iphone! Just wanted to say I love reading through your blog and look forward to all your posts! Keep up the outstanding work!
sKsQ
sKsQ,.).,”).(‘
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ’;SELECT/**/SLEEP(5)#
sKsQ’);SELECT/**/SLEEP(5)#
sKsQ’)));SELECT/**/SLEEP(5)#
sKsQ%’;SELECT/**/SLEEP(5)#
sKsQ”));SELECT/**/SLEEP(5)#
sKsQ);SELECT/**/SLEEP(5)#
sKsQ));SELECT/**/SLEEP(5)#
sKsQ)));SELECT/**/SLEEP(5)#
sKsQ;SELECT/**/SLEEP(5)#
sKsQ’;SELECT/**/SLEEP(5)–/**/INdN
sKsQ’)));SELECT/**/SLEEP(5)/**/AND/**/(((‘ADLX’=’ADLX
sKsQ’;SELECT/**/SLEEP(5)/**/AND/**/’LaNa’=’LaNa
sKsQ’);SELECT/**/SLEEP(5)/**/AND/**/(‘Tove’/**/LIKE/**/’Tove
sKsQ’;SELECT/**/SLEEP(5)/**/AND/**/’CYMP’/**/LIKE/**/’CYMP
sKsQ”);SELECT/**/SLEEP(5)/**/AND/**/(“OxeT”=”OxeT
sKsQ”);SELECT/**/SLEEP(5)/**/AND/**/(“tzwC”/**/LIKE/**/”tzwC
sKsQ”;SELECT/**/SLEEP(5)/**/AND/**/”Eigz”/**/LIKE/**/”Eigz
sKsQ’;SELECT/**/SLEEP(5)/**/OR/**/’nLpf’=’qrVd
sKsQ);SELECT/**/SLEEP(5)–/**/aTNy
sKsQ)));SELECT/**/SLEEP(5)/**/AND/**/(((6433/**/BETWEEN/**/6433/**/AND/**/6433
sKsQ;SELECT/**/SLEEP(5)
sKsQ’));(SELECT/**/*/**/FROM/**/(SELECT(SLEEP(5)))mjph)#
sKsQ”;(SELECT/**/*/**/FROM/**/(SELECT(SLEEP(5)))mjph)#
sKsQ);(SELECT/**/*/**/FROM/**/(SELECT(SLEEP(5)))mjph)#
sKsQ));(SELECT/**/*/**/FROM/**/(SELECT(SLEEP(5)))mjph)#
sKsQ;(SELECT/**/*/**/FROM/**/(SELECT(SLEEP(5)))mjph)#
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
sKsQ
Pretty component to content. I just stumbled upon your blog and in accession capital to claim that I acquire in fact enjoyed account your blog posts.
Anyway I’ll be subscribing for your feeds or even I success you
access persistently rapidly.
Sessão boa é aquela que termina no verde e com plano cumprido.
What i do not realize is actually how you are not really much more well-liked than you may be right now. You’re very intelligent. You realize thus considerably relating to this subject, made me personally consider it from numerous varied angles. Its like women and men aren’t fascinated unless it is one thing to do with Lady gaga! Your own stuffs great. Always maintain it up!
naturally like your website but you need to check the spelling on several of your posts. Many of them are rife with spelling problems and I find it very troublesome to tell the truth nevertheless I will certainly come back again.
Hello my family member! I want to say that this article is amazing, great written and come with almost all important infos. I would like to see extra posts like this.
Only a smiling visitant here to share the love (:, btw great pattern.
Today, I went to the beachfront with my children. I found a sea shell and gave it to my 4 year old daughter and said “You can hear the ocean if you put this to your ear.” She put the shell to her ear and screamed. There was a hermit crab inside and it pinched her ear. She never wants to go back! LoL I know this is completely off topic but I had to tell someone!
?Levantemos nuestros brindis por cada cazador de tesoros !
–
?Que la fortuna avance contigo con hazanas inolvidables exitos memorables !